Introduction
Surgical management of patients with extensive thoracic aortic disease can be performed in a 1- or 2-stage fashion.1-5 One stage repair involves either a clamshell thoracotomy with simultaneous replacement of the ascending aorta, transverse arch and descending aorta with a prosthetic graft1, or ascending aorta and arch replacement with simultaneous antegrade or retrograde stenting of the descending aorta.2-4 The E®-vita open prosthesis (Jotec GmbH, Hechingen, Germany) is a combined stent-prosthetic graft designed to facilitate 1-stage treatment of patients with extensive aortic aneurysm or aortic dissection.5 It can be performed without intraoperative angiographic or echocardiographic assistance.
Patient Selection and Preparation
 |
|
 |
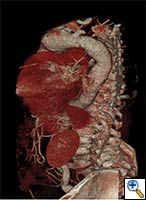 |
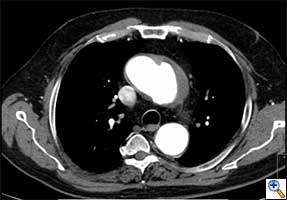
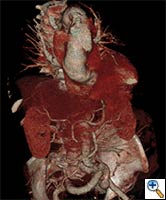
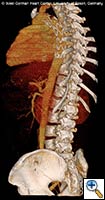
| FIGURE 1-3: 2D and 3D preoperative CT Images of a 78 year old man with aortic insufficiency, and ascending, arch and descending aortic aneurysm with mural thrombus. | |
Appropriate patients for this procedure have extensive thoracic aortic disease and a suitable landing zone for a stent graft in the descending aorta. Many of these patients are candidates for a 2-stage procedure, and selection is based on expected outcomes and the estimated interval rupture risk if patients were to be treated in a staged fashion. (see Figs. 1-3)
In addition to a comprehensive history, physical examination and laboratory parameters, our standard evaluation of these patients includes:
- Thorax CT angiogram with 3-D reconstruction, which is invaluable in the assessment of these patients. Attention must be paid to the detailed arch anatomy, taking note of any anomalies (eg. anomalous vertebral artery, bovine aortic arch), the diameter of the aorta distal to the LSCA, extent and diameter of the descending aorta aneurysm, and the diameter of the expected landing zone. Furthermore, the extent of atheroma/aortic calcification should be evaluated.
- Cardiac catheterization to exclude concomitant coronary artery disease or to evaluate patency of prior bypass grafts.
- Echocardiography to evaluate ventricular function as well as to exclude concomitant valvular pathology.
- Pulmonary function tests with appropriate optimization of pulmonary function, particularly since respiratory problems are a significant cause of morbidity in these patients.
- Carotid ultrasound to exclude significant carotid disease that may warrant simultaneous or staged endarterectomy or stenting. Carotid-subclavian bypass is occassionally necessary for these patients, depending on the arch anatomy and if coverage of the left subclavian artery (LSCA) is anticipated.
- CT scan of the brain may also be useful in some patients, particularly those with a history of recent cerebrovascular accident or neurologic deficits
Operative Procedure
- A complete median sternotomy is performed. Arterial cannulation is achieved via the right axillary artery (directly or via a 6-8 mm Dacron prosthesis), facilitating selective antegrade cerebral5perfusion during distal arrest if arch reconstruction is performed. Standard venous cannulation is performed via the right atrium.
- After establishment of cardiopulmonary bypass (CPB), the patient is typically cooled to 22-28ºC (bladder or rectal), depending on the complexity of the anticipated procedure.
- The innominate vein is encircled with a vessel loop and the arch vessels are separately encircled and ensnared with vessel loops or tourniquets. The innominate vein should be divided only when visualization of the aortic arch would be otherwise compromised.
- Depending on any accompanying aortic root or cardiac procedures, an antegrade cardioplegia cannula is placed and a LV vent is also inserted as the heart fibrillates.
- If a concomitant cardiac operation (CABG, valve replacement/repair, etc) is indicated, the aorta is clamped, the heart arrested, and this procedure is performed.
- Once the patient has been cooled to the desired temperature, the CPB flow is reduced to 500 ml/min, the innominate (brachiocephalic) artery is clamped or controlled, and the aortic arch is opened longitudinally. Complete circulatory arrest may be briefly required in order to fully assess the arch anatomy / pathology.
- An antegrade cerebral perfusion cannula (14, 17, or 20 Fr) is advanced into the left carotid artery. Bilateral selective antegrade brain perfusion (18oC) is initiated via a Y-connection from the arterial line to the left carotid artery cannula and flow is increased to 10-15 ml/kg/min to maintain a mean radial artery pressure of 50mmHg. The left subclavian artery is typically blocked with a 6-F Fogarty catheter (or snared) to prevent a steal phenomenon.
- During this period of lower body circulatory arrest, the size of the proximal descending aorta (ie. proximal anastomotic site) is confirmed before the E®-vita stent-graft prosthesis is opened. A stent-graft prosthesis that is 10% larger than the distal landing zone (as measured on preoperative CT) may be acceptable, but oversizing is often avoided, particularly in aortic dissection.
 |
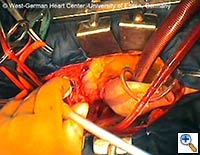 |
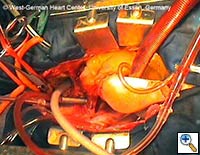 |
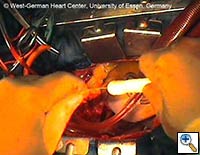
| Figures 4–6. These figures demonstrate advancement of the prosthesis over a transfemorally placed guide wire, the most secure method to assure appropriate placement. | ||
- The E®-vita prosthesis is then inserted into the descending aorta, with or without the accompanying guidewire. In particularly difficult descending aortic anatomy, a guidewire may be passed transfemorally, preferably via a previously inserted sheath. In some centers, the guide wire is a mandatory adjunct to secure the graft into the true lumen for aortic dissection and to avoid thrombus mobilization for aneurysms. (see Figs. 4-6 for placement over guide wire)
- The E®-vita prosthesis has 3 settings: P (Park), D (Drive), and N (Neutral). When initially unpacked, it is in Park mode. Following insertion into the descending aorta and stabilization, the mode is switched to Drive. The lever at the handle is then squeezed together repeatedly, which results in deployment of the stent. The surgeon must ensure that he keeps the position of his left hand, which is holding the lever portion of the deployment device, absolutely stable and does not pull the device back during stent deployment. Self-expansion of the stent is achieved by the nitinol wire skeleton.
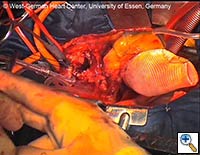
- The delivery system may then be passed off, and the prosthetic graft should be pulled only a few centimeters out of the stent-graft portion of the E®-vita prosthesis. The prosthetic graft is then sewn to the proximal descending aorta using a continuous polypropylene suture (preferably 3-0 Prolene with a “Visiblack” needle). This suture line should be performed 1 - 5 cm proximal to the stent-graft / prosthetic graft junction. (see Fig. 7)
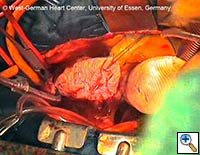
- The remaining portion of the prosthetic graft is then withdrawn out of the stent graft using the attached ligature. (see Fig. 8) Head vessel reimplantation is then performed according to anatomy and surgeon preference. We attempt to reimplant the LSCA whenever possible, usually as a separate graft during rewarming. The innominate artery clamp or tourniquet is released, and flow via the right axillary artery is slowly increased to deair the head vessels and the prosthetic graft. Once deairing is completed, the prosthetic graft is clamped, and full perfusion and rewarming is performed.
- During rewarming, any remaining cardiac procedures are completed, and the prosthetic graft is either sewn directly to the supracoronary ascending aorta or to a separate ascending aortic prosthesis.
- The heart is then deaired in standard fashion, and the aortic cross-clamp is removed. Once the patient has been rewarmed (typically 35-36 C bladder), the patient is weaned from CPB.
- Chest drains and pacing wires are placed, and a standard closure is performed.
- We recommend postoperative imaging to assess the quality of the aortic reconstruction. (see Fig. 9) Followup evaluation is particularly important to assess false lumen and branch vessel status in aortic dissection, and to monitor for interval progression of associated thoracoabdominal aneurysm disease.
Preference Card
Standard surgical set for cardiac/aortic procedures
E®-vita open prosthesis (Jotec GmbH, Hechingen, Germany) – This product is not FDA approved for use in the USA
Technical specifications:
- Hybrid polyester stent-graft prosthesis
- Stent graft portion available in diameters 24-40 mm
- Length 150 mm for diameters 24-30 mm
- Length 160 mm for diameters 33-40 mm
- Guidewire within pre-packaged delivery system
- Length 130 mm recently available
Tips and Pitfalls
- Study the preoperative CT carefully to develop a comprehensive operative plan that minimizes the risk of stroke and paraplegia. Identify abnormal arch anatomy, particularly anomalous subclavian or vertebral arteries.
- Keep in mind that a pre-, intra- or post-operative carotid-subclavian bypass allows for safe coverage of the LSCA. This procedure not only allows the stent graft to be placed more proximally, but reduces the risk of paraplegia, particularly in patients with prior abdominal aortic procedures.
- Avoid aggressive oversizing of the E®-vita stent-graft in order to reduce the risk of an aorto-esopahgeal fistula, a late complication after transfemoral or antegrade thoracic aortic stenting.
- Particularly when used in the setting of acute type A aortic dissection, angioscopy may be a useful adjunct to visualize the descending aorta for re-entry tears with subsequent deployment of the hybrid prosthesis. Post-deployment, repeat angioscopy can be used to evaluate unfolding and if needed, a post-dilatation with a balloon-tip catheter may be performed.
- A transfemorally inserted guidewire can also be a useful adjunct in selected cases, if not used routinely.
- Near infrared spectroscopy (NIRS) to monitor cerebral oxygen saturation is sometimes helpful to detect clinically relevant problems with cannulae and/or cerebral perfusion.
- Moderate hypothermia with bilateral selective antegrade cerebral perfusion is usually preferable to deep hypothermia with complete circulatory arrest. However,this technique may be associated with suboptimal protection of the spinal cord. If a prolonged period of circulatory arrest is anticipated, then deep hypothermia is preferable.
- Lower body perfusion via a femoral arterial cannula and clamping of the graft portion of the hybrid prosthesis after completion of the distal suture line is an useful adjunct to minimize the visceral/peripheral ischemic time.
Complications
Intraoperative complications
- Inadequate deployment of the stent graft
- Rupture of the descending aorta
- Iatrogenic descending aortic dissection
- Initiation or exacerbation of malperfusion during type A dissection
Postoperative complications
- Bleeding
- Stroke
- Paraplegia
- Respiratory Failure
- Renal Failure
- Aorto-esophageal fistula (late)
Postoperative Care
As with conventional arch and descending aortic procedures, careful attention must be paid to postoperative:
- Bleeding with expeditious replacement of clotting factors, and mediastinal reexploration as surgically indicated
- Assessment of neurologic status, in particular paraplegia, with timely insertion of a CSF drain and induced hypertension to minimize the risk of delayed paraplegia. Suspicion of cerebral deficits warrants an emergent CT scan. MRI is useful for diagnosis of spinal cord injury.
- Respiratory status, particularly since respiratory failure is a frequently encountered complication. Aggressive pulmonary therapy (e.g. repeated left bronchoscopy) and early tracheostomy may be indicated to reduce morbidity.
- Visceral ischemia, particularly in patients with aortic dissection, since some degree of baseline ischemia may be exacerbated by the procedure. Timely laparotomy and bowel resection may prove lifesaving.
- Renal failure, particularly in aortic dissection and compounded by preoperative shock.
Results
The first results of an international registry of E®-vita open patients were recently published. Of the 106 patients with aortic dissection (55 acute, 51 chronic) that were treated, successful stent graft deployment was performed in 99%, hospital mortality was 12%, and new stroke occurred in 5%. Subsequent false lumen thrombosis in the thoracic aorta occurred in 93% by the first follow-up CT examination.
Disclosure
Prof. Jakob serves as a consultant to Jotec GmbH.
References
- Kouchoukos NT, Masetti P, Mauney MC et al. One-stage repair of extensive aortic dissection using the arch-first technique and bilateral anterior thoracotomy. Ann Thorac Surg 2008;86:1502-9.
- Svensson LG, Kim KH, Blackstone EH et al. Elephant trunk procedure: newer indications and uses. Ann Thorac Surg. 2004;78(1):109-16.
- Etz CD, Plestis KA, Kari FA et al. Staged repair of thoracic and thoracoabdominal aortic aneurysms using the elephant trunk technique: a consecutive series of 215 first stage and 120 complete repairs. Eur J Cardiothorac Surg 2008;34(3):605-14.
- Pochettino A, Brinkman WT, Moeller P et al. Antegrade thoracic stent grafting during repair of acute DeBakey I dissection prevents development of thoracoabdominal aortic aneurysms. Ann Thorac Surg 2009;88(2):482-9.
- Tsagakis K, et al. Arch replacement and downstream stent grafting in complex aortic dissection: first results of an international registry. Eur J Cardiothorac Surg. 2011 Jan;39(1):87-93; discussion 93-4. Epub 2010 Jun 2.
- Roselli EE, Soltesz EG, Mastracci T et al. Antegrade delivery of stent grafts to treat complex thoracic aortic disease. Ann Thorac Surg 2010;90:539-46.





Comments