ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Bronchial and Pulmonary Arterial Sleeve Resection
Patient Selection
The indication for a sleeve resection for lung cancer is well established: a tumor arising at the origin of a lobar bronchus precluding simple lobectomy, but not infiltrating as far as to require pneumonectomy. Bronchial sleeve lobectomy is reported to be adequate for 5% to 8% of patients with resectable lung cancer but rates as high as 13% have been reported recently. It is important to point out that this increased rate of sleeve lobectomy is achieved at the expense of a decreased incidence of pneumonectomy and not of lobectomy, while the oncologic results remain unchanged.
From a functional point of view, sleeve lobectomy is strictly indicated in patients who cannot withstand pneumonectomy, but recent experiences have shown that the advantages of sparing one lung lobe are evident also in patients without cardio-pulmonary impairment.
Oncologically, the primary goal of surgery is complete resection of lung cancer with adequate resection margins free of tumor. This is all the more true for carcinoid tumors or benign lesions. Evidence has been obtained that there is little if any gain in extending the resection as far as pneumonectomy. These considerations apply also to patients with nodal involvement limited to hilar lymph nodes (N1).
Reconstructive surgery of the pulmonary artery has exactly the same indications, although this operation has been less frequently performed to date.
Operative Steps
Lobar sleeve resection
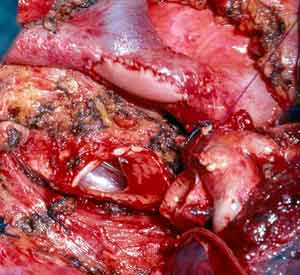
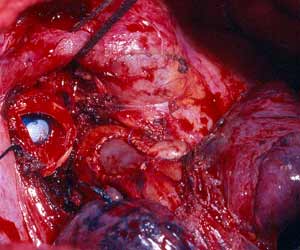
The first case illustrates a right upper lobe sleeve resection. The patient has already had patch reconstruction of the SVC (Figure 2). The azygos vein arch has been resected and the ends are ligated with silk. The right upper lobe has been resected, and a clear distal margin on the bronchus intermedius and proximal margin on the trachea have been obtained. It is important that the suture line is tension-free. This can be achieved by dividing the pulmonary ligament and, more often on the right side, by incising the pericardium around the inferior pulmonary vein.
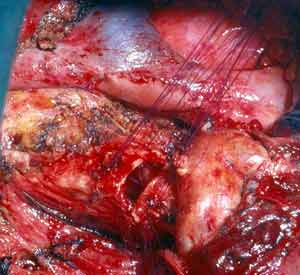 |
The bronchial anastomosis is performed with interrupted monofilament absorbable 4/0 sutures placed extramucosally on the cartilaginous portion. On the membranous portion, placing the sutures submucosally is not necessary because the sutures do not bulge in the bronchial lumen if they are properly tied. Initially, two sutures are placed on the far (mediastinal) end of the cartilaginous portion and are tied extraluminally. The surgeon ties the suture on his side while the assistant approximates his to relieve tension. Subsequently, the assistant ties his suture without tension thanks to the previously tied suture. The membranous portion sutures are then placed and tied (Figure 3).
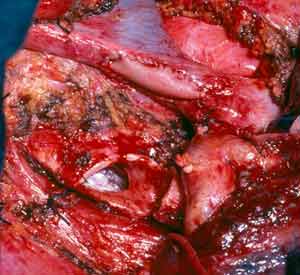
The remaining sutures are then placed on the rest of the bronchial circumference and are left untied (Figure 4). The sutures are then tied, starting from the either end of the cartilaginous portion and working towards the middle. Placing and tying the sutures in this order allows compensation for even large caliber discrepancies. This technique prevents torsion of the bronchial axis and gently stretches and dilates the circumference of the distal bronchus. The larger bronchial stump works as a stent, increasing the caliber of the anastomosis and minimizing secretion retention in the early postoperative course when edema at the site of the anastomosis is more likely to occur. The anastomosis is wrapped with a vascularized pedicle of autologous tissue, usually an intercostal muscle flap.
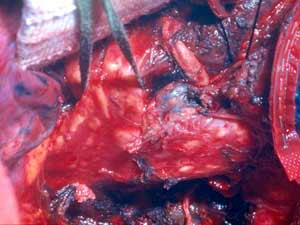 |
 |
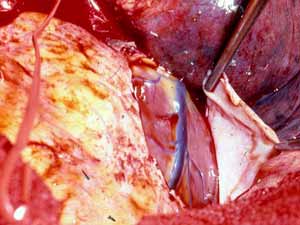
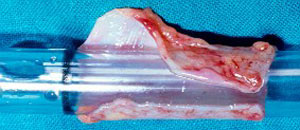
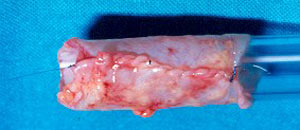
| Figure 5: The right upper lobe bronchus has been preserved for reimplantation. Its short length before (left) and after (right) division is evident. | |
"Y"resection with lobar reimplantation
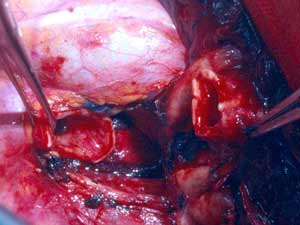
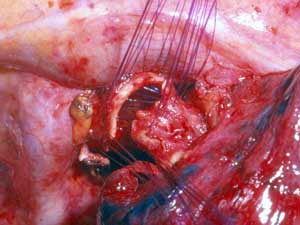
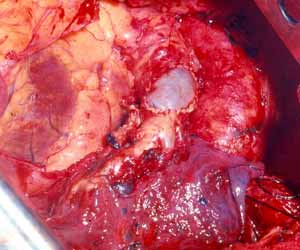
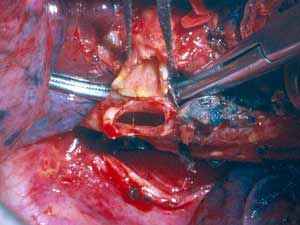
In case of upper lobe reimplantation after “Y” sleeve resection, a few technical points should be considered. The upper lobe bronchus is often so short that it is reduced to a disk of bronchial tissue where the openings of the segmental bronchi can be seen. This is more frequent on the right side (Figure 5). Also, the pulmonary artery and the lung are very close to the suture line and the exposure is poor, especially on the mediastinal side of the anastomosis (Figure 6). The anastomosis is performed in the same fashion as described above (Figure 7).
 |
Patch reconstruction of pulmonary artery
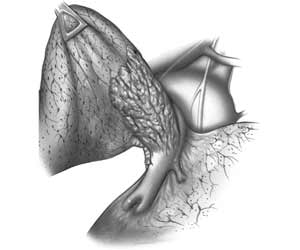
This technique is very versatile and can be used in a variety of circumstances. These range from limited infiltration involving the origin of segmental arteries to large defects extended longitudinally on the PA (Figures 8, 9). The only necessary condition is that the opposite side of the circumference of the PA is free from tumor.
 |
 |
| Figure 9: Line drawing (left) and intraoperative image (right) showing the usual pattern of infiltration of the interlobar portion of the pulmonary artery posterior to the left upper lobe bronchus.* | |
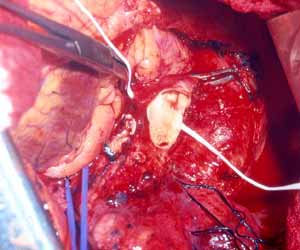 |
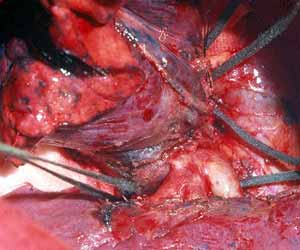
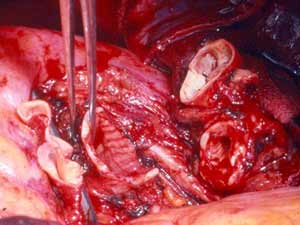
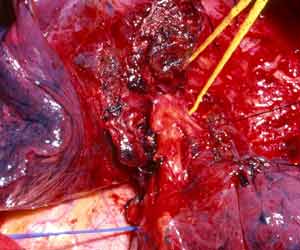
During resection control of the main pulmonary artery and the inferior pulmonary vein is obtained. The superior pulmonary vein is divided in a standard fashion. Pulmonary arterial tissue is resected en bloc with the specimen. After the resection, an oval defect oriented along the PA axis remains, even if the resected portion was circular in shape (Figure 10). This is due to the tension applied on the vessel by the lower lobe. The patch should be tailored according to the size and shape of the resected portion rather than according to the PA defect (Figure 11). After the patch is secured to the artery by 5/0 or 6/0 monofilament running suture, the PA is declamped (Figure 12). The suture line must be checked carefully for oozing, which might not be evident due to the low PA pressure. Also it is important to check the position of the artery after reexpansion of the lower lobe, for kinking might occur.
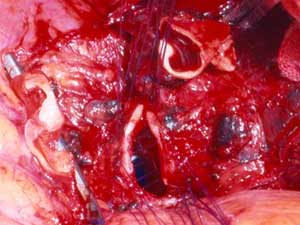
Infiltration of the PA on the right side requiring arterial reconstruction is less frequent. Partial resection of the PA often is performed in conjunction with right upper lobe sleeve resection (Figure 13). After right upper sleeve lobectomy en bloc with partial resection of the PA, the stump of the main bronchus and the PA defect are left open in the field (Figure 14). Harvesting of the pericardium is performed as above, based on the size of the portion of the resected pulmonary artery. Patch reconstruction of the PA is completed as above before reimplantation of the bronchus to reduce the arterial clamping time (Figure 15).
 |
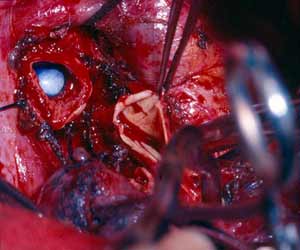
On occasion a sleeve resection of the upper lobe and the pulmonary artery is necessary (Figure 16). Pulmonary artery and inferior pulmonary vein control are obtained as described above. The vascular conduit is constructed from a segment of autologous pericardium (Figure 17). When sizing the conduit, two points must be considered: the PA stumps can be approximated closer than it seems, and the conduit will stretch more than predicted. It is advisable to tailor the length of the conduit on the basis of the resected arterial segment, because the elasticity of the two tissues is comparable. The bronchial anastomosis is performed first to avoid traumatizing the PA (Figure 18). The PA is reconstructed by end-to-end anastomosis with running 5/0 or 6/0 monofilament suture (Figure 19).
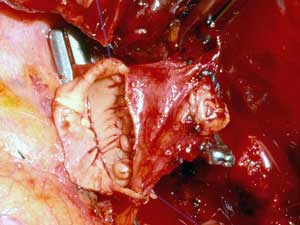 |
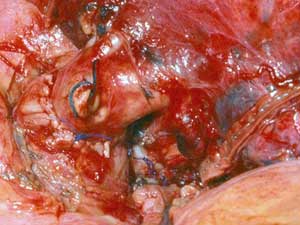 |
| Figure 19: The arterial conduit is interposed (left) and the proximal and distal anastomoses have been completed (right). | |
*Figure 9 Reprinted with permission of Rendina EA and Venuta F "Reconstruction of the Pulmonary Artery," Chapter 36 in Pearson's Thoracic Surgery, Philadelphia, W.B. Saunders, 2002.
Preference Card
- 2.5 magnification loupes
- Headlight
- Long (24cm) heavy titanium Castro-Viejo needle holder (for PA sutures)
- Long (24cm) fine tooth forceps (for manipulating the bronchus)
Tips & Pitfalls
- It is important that in candidates for a sleeve resection preoperative bronchoscopy is performed by one of the operating surgeons. This is advantageous at the time of the operation, when the bronchi are incised and divided. It is also useful to have precise knowledge of the preoperative and intraoperative appearance of the airway if any bronchial complication should occur and laser recanalization or stenting should become necessary. Bronchoscopy is performed under local anesthesia to observe bronchial motion during voluntary breathing and coughing, and multiple biopsies are taken. Careful evaluation of bronchial motion is important to infer the state of tissues outside the bronchus, for stiffness of the bronchial wall may indicate peribronchial tumor infiltration. This is particularly important in areas where the bronchus is known to be adjacent to the PA, which might consequently be involved.
- Sometimes the distal bronchial incision falls close to the takeoff of the superior segmental bronchus or middle lobe bronchus. This is not a contraindication to the operation, but care must be taken to avoid stricture of the tiny segmental bronchi falling close to the suture line.
- The use of steroids in the perioperative period in patients undergoing tracheobronchial resection is controversial. We believe that the antiedema effect of steroids is beneficial because it reduces secretion retention and atelectasis, it facilitates parenchymal reexpansion, and it minimizes the risk of dehiscence and granuloma formation. Aerosolized steroids (methylprednisolone 5mg twice a day) are also part of our preoperative treatment when sleeve lobectomy can be predicted before hand. It is our experience that patients treated with steroids do not need bronchoscopy or close observation in the postoperative period. They recover faster and leave the hospital earlier.
- When the sleeve resection is planned preoperatively, we prepare an intercostal pedicle flap before opening the chest to avoid crushing the intercostal vascular bundle. The intercostal muscle flap is precious for protecting the anastomosis, and we use it routinely in all our bronchial reconstructions. The flap affords additional protection against anastomotic failure and preserves the continuity of the airway in case of small dehiscence, thus avoiding bronchopleural fistulas. The risk of erosion of the adjacent PA is also minimized. In addition, the revascularization of the distal bronchus avoids problems related to ischemia.
- The postoperative course of bronchial sleeve resection depends to some extent on patient compliance and judicious clinical management. The short-term results of PA reconstruction depend mostly on operative judgment and technique. If the operation has been correctly performed, specific complications may be expected in no more than 5% of the patients.
- Because the PA is a low pressure vessel, leakage from the suture line may go unnoticed intraoperatively. Also, the bleeding may start in the first or second postoperative day after a patch reconstruction. A blood loss of up to 800ml to 1000ml daily may occur after 1 or 2 days of no drainage. This may last for 1 or 2 days and then stop spontaneously independent of anticoagulant usage. A possible explanation is that the autologous pericardium shrinks and curls markedly after harvesting, and it is difficult to place the suture bites at the appropriate distance. After declamping and distention, bites too wide apart may result. These would not cause bleeding immediately because the PA is stretched downward by the atelectatic lower lobe, and simple apposition of the tissue edges is enough to overcome the low PA pressure. However, in the postoperative period, when the reexpansion of the lower lobe elevates the hilum, the rotation and kinking of the PA may distort the suture line and open a bleeding site. It is therefore very important, especially when using autologous pericardial patches, to carefully check the suture line and test the PA position after reexpansion of the residual lobe. The latter maneuver is also important to prevent thrombosis. After a patch reconstruction of the PA associated with a bronchial sleeve, the bronchial axis is shortened, and the length remains stationary. Some of the discrepancy is compensated by the elasticity of the vessel, but the PA may tend to kink and fold over itself. The aforementioned repositioning of the PA due to the reexpansion of the lower lobe further increases the risk. Impairment of blood flow may ensue, and thrombosis may be facilitated. Under these circumstances, it is better to cut the distorted segment away and proceed to an end-to-end anastomosis.
- Sometimes sleeve resection and end-to-end anastomosis are anatomically impossible, such as in cases of left upper lobe tumors infiltrating the concave surface of the PA from its origin down to the anterobasal artery. On the right side, the same problem may arise when the posterolateral aspect of the PA is infiltrated from the upper division artery to the artery for the superior segment of the lower lobe.
- Sleeve resection is sometime excessive if the artery is only partially infiltrated.
- An end-to-end anastomosis can be technically difficult, owing to unexpected traction between the stumps and caliber discrepancy. Tears on the arterial wall while suturing are difficult to repair, and failure to do so may produce disastrous results.
- The main pitfalls of the use of a conduit are its sizing and length. Application of the previously mentioned technical insights will prevent this problem.
Results
The recent literature contains a number of reports on the favorable results of sleeve resection. Compared to pneumonectomy, complications and mortality are lower, cardiopulmonary function is more satisfactory, and long-term survival rates are comparable. In addition, a number of papers dealing more specifically with resection of the pulmonary artery have been published during the last fiew years. A summary of the most recently published data is presented in Tables I to IV. All the quoted papers appear in the English literature and can easily been found in Medline.
TABLE I – Bronchial sleeve resection
| Author | Year | Patients | Complications (%) |
Technical Complications (%) |
Mortality (%) |
Survival (5 yr; %) |
| Kawahara | 1994 | 112 | ns | 15.6 | ns | ns |
| Van Schil | 1996 | 145 | 18.6 | ns | 4.8 | 46 |
| Gaissert | 1996 | 72 | 11 | 1.3 | 4 | 42 |
| Rea | 1997 | 217 | 12.5 | ns | 6.2 | 49 |
| Icard | 1999 | 110 | 50 | 4.5 | 2.75 | 39 |
| Kutlu | 1999 | 100 | 12 | 2 | 2 | 49* |
| Massard | 1999 | 63 | 28.5 | 9.5 | 1.6 | 43 |
| Suen | 1999 | 77 | 41.3 | 3.8 | 5.2 | 37.5 |
| Tronc | 2000 | 184 | 14.1 | 3.2 | 1.6 | 52 |
| Okada | 2000 | 151 | 10 | 0 | 0 | 48 |
| Lausberg | 2000 | 81 | ns | 0 | 1.2 | 61.9** |
| Rendina | 2000 | 145 | 12.4 | 2.7 | 3 | 37.9 |
| Hollaus | 2001 | 15 | 26.6 | 6.6 | 0 | 48*** |
| * From 6 to 168 months, **2 years, ***58 months | ||||||
TABLE II – Reconstruction of the PA alone or associated with bronchial sleeve
| Author | Year | Patients | Complications (%) |
TechnicalComplications (%) |
Mortality (%) |
Survival (5 yr; %) |
| Rendina | 1999 | 52 | 13.4 | 1.9 | 0 | 38.3 |
| Icard | 1999 | 16 | ns | ns | ns | 39* |
| Shrager | 2000 | 33 | 6.1 | ns | ns | 46.6 |
| Lausberg | 2000 | 4 | ns | 0 | ns | 61.9* |
| Okada | 2000 | 21 | ns | 0 | 0 | 48* |
| * Overall survival of PA and bronchial reconstruction | ||||||
TABLE III – Five and 10 year survival rates after bronchoplastic procedures by lymph node status
| Author | Year | Patients | 5 year survival (%) | 10 year survival (%) | ||||
| N0 | N1 | N2 | N0 | N1 | N2 | |||
| Rea | 1997 | 179 | 72 | 36 | 22 | 59 | 27 | 14 |
| Icard | 1999 | 110 | 57 | 29 | 33 | 26 | 18 | 33 |
| Tronc | 2000 | 184 | 63 | 48 | 6 | 48 | 27 | 0 |
| Van Schil | 2000 | 145 | 62 | 29 | 31 | 53 | 21 | 6 |
| Okada | 2000 | 60 | 70 | 21 | 55 | 0 | ||
TABLE IV – Comparative results of sleeve lobectomy and pneumonectomy in NSCLC
| Author | Year | Patients | Complications (%) | Mortality (%) | 5 year survival (%) | Local recurrence (%) | |||||
| Sl | Pn | Sl | Pn | Sl | Pn | Sl | Pn | Sl | Pn | ||
| Yoshino | 1997 | 29 | 29 | 13.7 | 24.1 | 0 | 6.9 | 65.7 | 58.8* | - | - |
| Suen | 1999 | 58 | 142 | 1.7 | 7.0 ** | 5.2 | 4.9 | 37.5 | 35.8 | - | - |
| Okada | 2000 | 60 | 60 | 13 | 22 | 0 | 2 | 48 | 36 | 8 | 10 |
| Sl : Sleeve Lobectomy Pn : Pneumonectomy * 3 year survival ** only postoperative respiratory failure |
|||||||||||
References
- Okada M, Yamagishi H, Stake S, Matsuoka H, Miyamoto Y, Yoshimura M, Tsubota N. Survival related to lymph node involvement in lung cancer after sleeve lobectomy compared with pneumonectomy. J Thorac Cardiovasc Surg 2000;119:814-9
- Rendina EA, De Giacomo T, Venuta F, Ciccone Am, Coloni GF. Lung conservation techniques: bronchial sleeve resection and reconstruction of the pulmonary artery. Semin Surg Oncol 2000;18:165-72
- Rendina EA, Venuta F, Degiacomo T, Ciccine AM, Moretti M, Ruvolo G, Coloni GF. Sleeve resection and prosthetic reconstruction of the pulmonary artery for lung cancer. Ann Thorac Surg 1999;68:995-1002
- Suen HC, Meyers BF, Gutrie T, Pohl MS, Sundaresan S, Roper CL, Cooper JD, Patterson GA. Favorable results after sleeve lobectomy or bronchoplasty for bronchial malignancies. Ann Thorac Surg 1999;67:1557-62
- Tronc F, Gregoire J, Rouleau J, Deslauriers J. Long term results of sleeve lobectomy for lung cancer. Eur J Cardio-Thorac Surg 2000;17:550-556
- Van Schil PE, Brutel de la Riviere A, Knaepen PJ, van Swieten HA, Reher SW, Goossens DJ, Vanderschueren RG, van den Bosch JM. Long term survival after bronchial sleeve resection: univariate and multivariate analyses. Ann Thorac Surg 1996;61:1087-91