ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Ligation of the Thoracic Duct for Chylothorax
Patient Selection
Most patients who have a high volume chylothorax and who require operative repair have an iatrogenic chylothorax. This most commonly occurs after esophagectomy. This is because the chylothorax is secondary to an injury to the main thoracic duct. The patient who has a chylothorax after a pulmonary resection rarely requires re-operation because the leak is usually from a tributary of the main duct as a result of lymph node dissection. For that reason, and since spontaneous chylothoraces (usually from lymphoma) rarely require thoracic duct ligation, this discussion is focused on the post-esophagectomy patient.
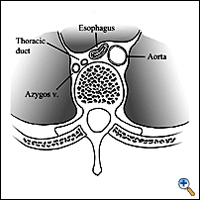
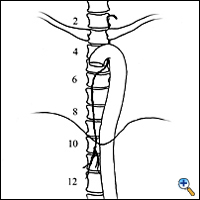
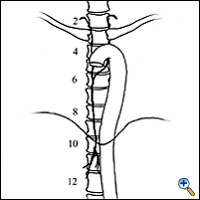
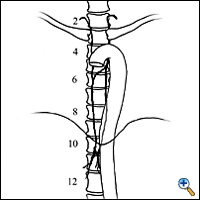
The thoracic duct is a tubular structure that is 2 to 3 mm in diameter, valved, and paper-thin. It is the main conduit of the lymphatic system. It is a fibrin-less system that runs in the posterior mediastinum. It originates as the cisterna chyli, which lies on top of the second lumbar vertebral body, and ascends anterior to the vertebral bodies, usually on the right side (Figure 1). It enters the chest through the aortic hiatus, crosses from the right side of the chest to the left at the level of the fourth or fifth thoracic vertebra, and usually empties into the left jugulosubclavian venous junction (Figure 2). It has a highly variable course (Figure 3) and is subject to injury during any abdominal, thoracic, or neck procedures in these areas. Trauma to the main duct rarely closes spontaneously.
Chylothorax after esophagectomy should be suspected when there is an unexplained high volume chest tube output that turns milky white after enteral tube feedings are started. It is often serosanginous until then. The diagnosis is confirmed by a triglyceride level of 110 mg/dl or greater, the presence of chylomicrons in the chest tube drainage, a positive Sudan stain, or, if needed, lipoprotein electrophoresis. Once the diagnosis is secured early intervention is critical. Although an exact volume of output as a threshold has not been shown, when the effluent is greater than 800 cc per day for 4 –5 consecutive days waiting for the leak to stop on its own is a waste of time. Moreover, it risks leukopenia and malnutrition. We suggest re-operation with thoracic duct ligation within five to seven days after the initial procedure. Total parental nutrition with complete cessation of all oral intake, somatostatin, medium chain triglyceride diets and percutaneous injection of sclerotic agents into the cisterna chyli will most likely fail in this situation. Re-operation should not be delayed.
Prior to re-operating a contrast swallow should be performed to ensure there is no large anastomotic leak that should also be addressed at re-operation. Some authors recommend a lymphangiogram prior to re-operation. This can be helpful in delineating the anatomy of the thoracic duct, which is aberrant in almost 40% of patients. The study can also identify the leak. However, lymphangiography is difficult to perform and few radiologists across the country do it enough to do it well. They have little experience in cannulating a small duct in the foot and then performing and reading the test.
Operative Steps
Once re-operation is decided upon, the first step is to decide where to make the incision. Lymphatic leaks can occur in the neck, in the abdomen, or in either or both chests, and they can be controlled in any of those compartments. This discussion focuses on transthoracic ligation, but the surgeon must decide which chest to enter. Usually the decision is simple – explore whichever chest has the chest tube in it with the chyle draining out of it. However, often after an Ivor Lewis operation the chyle can drain primarily into the left chest. There may be some drainage of chyle out of the right-sided chest tubes but the gastric conduit obscures a large pleural effusion. In this situation a left-sided tube should be placed and the amount of drainage should be measured in order to determine which approach is best. Re-operation usually is best via a re-do right thoracotomy. The main advantage of ligating the thoracic duct via a left VATS or left thoracotomy is that the conduit does not have to be retracted off the vertebral bodies and is not in the way. The disadvantages of going on the virginal left side are that you have committed the patient to recover from bilateral thoracotomies and you cannot perform a mechanical pleurodesis to help obliterate the pleural space.
Since a re-do right thoracotomy is most commonly needed and recommended we will discuss it step by step:
- The epidural should be left in, in preparation for the re-operation.
- A double lumen tube should be employed and is critical for either a VATS or a re-do thoracotomy to optimize exposure.
- A nasogastric tube should be left in or positioned with fluoroscopy into the gastric conduit prior to placing the patient in the left lateral decubitus position.
- Cream should be administered into the feeding jejunostomy (if one is not present it can be given through the nasogastric tube) just prior to prepping and draping.
- Re-open the chest.
- Drain all the effluent and try to pinpoint the site of the leak. The conduit can be easily mobilized off of the posterior mediastinum within 5 – 9 days, exposing the vertebral bodies. A white, milky jet can usually be seen at the level of the injured thoracic duct.
- The assistant should retract the conduit off the vertebral bodies. The vector of this retraction is important. It should be anterior (towards the sternum) more than medial (towards the heart). If the retraction is just medial towards the heart, potential stretch on the anastomosis and injury can occur. It also impedes inflow to the heart and may lead to decreased blood pressure or arrhythmias.
- The assistant must keep any retractor away from the right gastro-epiploic artery. This arcade is usually 180 degrees away from the lesser curve of the stomach that is the part of the conduit that is retracted anteriorly.
- Once the leak is clearly identified, ligation is carried out a few centimeters
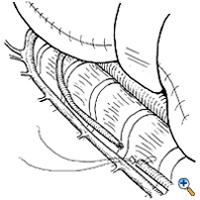
Figure 4. Technique for suturing a thoracic duct injury using pledgets for reinforcement. below it and also above it. Pledgeted 4-0 monofilament nonabsorbable sutures (Figure 4) are used to sew it closed both above and below the fistula. The leak should be completely stopped. Alternative methods to suturing the duct include clipping it, tying it, or ligating it cephalad only. These latter techniques require precise identification of the duct and one must be sure there are no small collaterals near the tear that will be missed with these methods. A dry gauze should be draped over the area to ensure it is dry. A new dry gauze pad should be left there while you perform the mechanical pleurodesis.
- A mechanical pleurodesis should be performed throughout the entire chest and especially on the diaphragmatic surface
- Recheck the area of the closure. It should still be dry. Remove the gauze pad and then spray or apply glue, FocalSeal® (Genzyme Biosurgical, Cambridge, MA), or another type of sealant over the area.
- Place three chest tubes, including two right angled tubes in the posterior and anterior aspects of the chest, just on top of the diaphragm but not touching the pericardium or the conduit.
- Close the chest.
Preference Card
No special equipment is needed. A wide malleable retractor is ideal to help retract the stomach up and off of the vertebral bodies. The FocalSeal® or other sealant should be prepared and the cream needs to be ordered pre-operatively.
Tips & Pitfalls
- Knowing when to re-operate on any of your own patients is difficult. But if a patient has had an esophagectomy and has greater than 800 cc per day of chylous effusion for 3 –4 consecutive days, re-operation is needed and should be done without delay.
- If you have performed a transhiatal esophagectomy, are sure the leak can be controlled from the abdomen without the need for a thoracotomy, and a chest tube has been placed and the chyle is coming out of the chest tube, perform a lymphangiogram prior to re-exploring the abdomen. If the leak is high in the chest, simple ligation of the main duct at the level of the hiatus via a laparotomy may not be sufficient and the fistulous effusion may continue. The patient may require a third operation.
- Do not give the cream via the jejunostomy tube too early. Give it about 20 to 30 minutes prior to induction of anesthesia to optimize visualization of the leak.
- Do not use methylene blue. It can stain the surrounding tissue and can make finding the leak difficult.
- Do not sew the duct without pledgets. The thoracic duct is paper-thin and chyle contains no fibrin. Non-pledgeted sutures will tear it further. Sew it above and below the leak and do not place the stitches through the duct but into surrounding tissue around the duct. Allow the pledgets to close the duct. Use of clips may be an adjunct, but I prefer pledgeted sutures.
- Do not settle for a small dribble. Keep sewing until it is bone dry. Then, even if it looks dry, add the "belt and suspenders" of topical glue and pleurodesis. These extra precautions add little time or morbidity. A failed procedure and the need for a third operation within 10 days will erode patient and family confidence in you.
- The assistant’s retraction is critical. Ensure that the anastomosis is not being stretched. Usually if an Ivor Lewis has been performed the anastomosis should be above the azygous vein and there still should be lots of redundancy in the conduit. The retraction should not pull much on the anastomosis.
Results
The results of early closure of the thoracic duct for a high output iatrogenic chylothorax after esophagectomy are excellent. When re-operation is not delayed and simple duct closure of any type is performed, patients have little added morbidity and the reported success rates are around 90%. The technique described above features not only duct ligation caudal to the leak but also includes ligation of the cephalad end of the duct, and adds a mechanical pleurodesis and sealant or glue to cover the area of the leak. When this technique is used the success rate approaches 100%. In our experience in 13 patients there have been no failures utilizing this technique. Moreover, ten of these 13 patients had long delays in resolution of chylothorax owing to either failed medical treatment or failed re-operative treatment performed at other institutions. If one performs a second re-operation on a persistent leak after an attempted repair, especially if the patient’s other operations were performed elsewhere, we strongly recommend a pre-operative lymphangiogram. It ensures that the duct does not have some unusual course and that no large leaking collateral is present.
References
- Cerfolio RJ, Allen MS. Postoperative chylothorax. J Thorac Cardiovasc Surg 1996;112:1361-6
- Miller JI Jr. Diagnosis and management of chylothorax. Chest Surg Clin N Am 1996;6:139-48
- Hillerdal G. Chylothorax and pseudochylothorax. Eur Respir J 1997;10:1157-62
- Mohlala ML, Burrows RC, Mokoena TR. Early operative management of chylothorax by thoracic duct ligation. S Afr J Surg 1989;27:11-2
- Vallieres E, Shamji FM, Todd TR. Postpneumonectomy chylothorax. Ann Thorac Surg 1993;55:1006-8
- Paes ML, Powell H. Chylothorax: an update. Br J Hosp Med 1994;51:482-90
- Johnstone DW, Feins RH. Chylothorax. Chest Surg Clin N Am 1994;4:617-28
- Sieczka EM, Harvey JC. Early thoracic duct ligation for postoperative chylothorax. J Surg Oncol 1996;61:56-60