Incidence and Significance
- Peripheral arterial disease may affect as many as 12 million people in the United States [1]. In the majority of patients the first symptom is intermittent claudication (IC). IC is characterized pain in one or both legs with walking, primarily the calves, but can also affect the thigh and buttocks. It resolves with rest. Approximately 1/3 of patients with IC will progress to critical limb ischemia, which is characterized by rest pain and/or tissue loss, about 1/5 of all patients with IC will require lower extremity revascularization over a 10-year period of follow up [2, 3]. IC should be considered part of a systemic vascular disorder, in that 60% of patients die from coronary artery disease, and 10% from stroke.
Medical Management
- A critical aspect of managing IC is risk factor adjustment and medical management. Risk factors assessment includes determining the presence of carotid and coronary stenosis, and in patients with a strong smoking history, screening for lung cancer.
- Management of hypercholesterolemia, hyperlipidemia, detecting homocystinemia, smoking cessation and careful foot care in diabetic patients are all critical and yet while helping with the systemic risk factors have minimal immediate impact of IC.
- Antiplatelet therapy has benefit both as medical management and after reconstruction. Aspirin has been shown to reduce the risk of cardiovascular mortality in patients at risk [4, 5]. Low dose ASA (75-150 mg/day) appears to be as effective as high dose (500-1500 mg/day) with decreased incidence of GI bleed. Addition of Dypyridamole does not appear to have additional benefit. Ticlopidine and Clopidogrel, which inhibit platelet ADP-induced aggregation and activation, are also effective, although the former has a 1/2000-1/4000 incidence of TTP, neutropenia and/or thrombocytopenia. The CAPRIE trial compared clopidogrel (Plavix) 75 mg/day vs. ASA 375 mg/day. Primary endpoints included MI, stroke or other vascular death. There was an overall risk reduction of 8.7% (p<0.05) favoring clopidogrel. Of the 19000 patients, 6452 had a history of IC with either prior bypass or ABI’s < 0.85. There was an overall 23.8% reduction (ASA events/year 4.86% vs. clopidogrel 3.7%, p=0.0028) favoring clopidogrel, with lower GI side effects with clopidogrel [6, 7]. There was an overlap in confidence limits which means that the true difference could not be reliably estimated. Intriguingly, ASA appeared to have a better effect in incidence of MI and an ongoing study suggests that the combination may be better in patients with acute coronary syndromes, although the benefit of combination in PAD has not been absolutely established [1, 8].
Vascular Lab
- Vascular lab assessment, in conjunction with the clinical picture, can be invaluable. Plethysmography, which detects volume changes and records wave forms, is probably the best form of assessing patients with non-compressible vessels. Doppler, including imaging and pressure recordings, are also useful in determining prognosis and location of lesions.
|
Ankle-Brachial Index (ABI) |
Clinical Features |
|
1.1 + 0.1 |
Normal |
|
0.6 + 0.2 |
Claudication |
|
0.3 + 0.1 |
Ischemic rest pain |
|
0.1 + 0.1 |
Impending tissue loss |
- Exercise testing can reveal “occult” disease. A drop of 20% or 0.2 ABI with 5 minute or less treadmill confirms the diagnosis. The Duplex velocity patterns can also be used to assess lesions (including post-operatively).
|
Degree of Stenosis |
Waveform |
|
0% |
Normal |
|
1-19% |
Normal waveform, spectral broadening |
|
20-49% |
Marked broadening. 30% increase in peak systolic velocity |
|
50-99% |
Marked broadening, 100% increase in peak systolic velocity, loss of reverse diastolic flow component |
|
Occluded |
No detectable flow signal |
- While in as many as 90% of cases, lower extremity revascularization can be accomplished based on the information obtained from the vascular lab alone, a limiting factor is evaluation of the distal anastamotic site. For this reason, many prefer to obtain angiogram prior to operation.
- Following vein bypass, 20-30% will develop re-stenosis. 80% of recurrent occlusions following intervention develop in the first year, but the patients should be evaluated for life, to detect early, and therefore more easily corrected, lesions.
- Following revascularization (by any method), serial vascular lab assessment is recommended every 3 months for the first year and then every 6 months. The ratio of the peak systolic velocity just proximal to an anastamosis is compared to points distal. A PSV ratio of > 4 or > 300cm/s indicates a critical graft stenosis, and intervention should be considered. PSV ratios 2-4 indicate need for re-evaluation in 3 months.
Indications for Intervention
- There have been attempts to classify symptoms to provide some basis for recommending intervention. The Rutherford systems distinguishes between category 1 (mild), 2 (moderate) and 3 (severe) [9].
- The Fontaine classification defines stage IIa as walking more than 200m, and IIb as ability to walk less than 200m [10]. The Fontaine IIb category includes both the Rutherford stages 2 and 3.
- Traditionally, the indications for surgical intervention have been major clinical symptoms, such as rest pain, non-healing ulceration or at least severe claudication. It has been argued that, as interventional approaches usually are associated (at least acutely) with lower risk than operative approaches, these should be considered earlier. On the other hand, any proposed intervention has a concomitant requirement to be associated with minimal risk, including not complicating a later surgical bypass or reconstruction. As interventional approaches are being used more commonly on more complex lesions, driven by patient risk factors for surgery, the concept of endovascular-first as a strategy is gaining ground, as long as surgical approaches are not summarily precluded and the anatomy of the lesion to be treated does not suggest a higher risk of embolization, rupture dissection, etc. [1].
- Most up-to-date (modified) classification for femoral popliteal disease has been designed by the TASC II group consisting of multi-disciplinary international group of physicians, surgeons and interventionalists (Table 1) [14]. According to the TASC II consensus statement, recommendations for endovascular vs. open revascularization for femoral popliteal disease are the following:
Endovascular therapy is the treatment of choice for type A lesions and surgery is the treatment of choice for type D lesions. Furthermore, endovascular treatment is the preferred treatment for type B lesions and surgery is the preferred treatment for good-risk patients with type C lesions. Further, treatment recommendations for type B and type C lesions are based on the patient’s co-morbidities, fully informed patient preference and the local operator’s long-term success rates.
Table 1. TASC Classification of Femoral Popliteal Lesions.
Type A lesions _ Single stenosis ≤10 cm in length.
_ Single occlusion ≤5 cm in length.
Type B lesions _ Multiple lesions (stenoses or occlusions), each ≤5 cm.
_ Single stenosis or occlusion ≤15 cm not involving
the infra geniculate popliteal artery.
_ Single or multiple lesions in the absence of continuous
tibial vessels to improve inflow for a distal bypass.
_ Heavily calcified occlusion ≤5 cm in length.
_ Single popliteal stenosis.
Type C lesions _ Multiple stenoses or occlusions totaling >15 cm with
or without heavy calcification.
_ Recurrent stenoses or occlusions that need treatment
after two endovascular interventions.
Type D lesions _ Chronic total occlusions of CFA or SFA (>20 cm,
involving the popliteal artery).
_ Chronic total occlusion of popliteal artery and
proximal trifurcation vessels.
Access
- There are four potential approaches for obtaining access, the choice of which depends on specific anatomy and patient body habitus.
- Lesions that involve the origin of the superficial femoral artery (SFA) can be approached by a combined open cut down (possibly in conjunction with a patch angioplasty) and simultaneous more distal intervention. These lesions usually cannot be simply stented as the origin of the profunda femoral artery (PFA) will be encroached upon, and in many, if not the majority, the PFA represents a critical source of collateral flow. When performing an open exposure, in general a longitudinal incision is used which, although associated with increased risk of lymph leak and slightly greater wound healing issues, provides more flexibility in exposure than an oblique incision. If pre-operative workup suggests that the disease is focal and the need for more proximal control is minimal, than we do favor an oblique incision. Advantages of the combined open-endovascular approach is the following: a patch angioplasty reliably opens the origin of the SFA without compromising the PFA; endartectomy of the CFA and PFA can be performed; the intervention zone for stenting is moved distally, such that a stent can be placed at the time or subsequently that does not overlie the origin of the PFA; there is greater ease in accessing the SFA in an atraumatic manner; if it is not possible to cross the SFA/PA then the option remains at the same setting to perform surgical bypass; if there are concomitant proximal iliac lesions, these can be easily addressed in the same setting. The general strategy is to perform the endartectomy and patch angioplasty and then through the roof of the patch, perform subsequent interventions.
- Lesions that involve a segment within 0.5-1.0 cm of the origin of the SFA can be approached by either the crossover technique from the contralateral CFA or from a retrograde popliteal artery (PA) access. These can be combined using a snare to obtain through-and-through access which provides greater stability. We believe that careful use of ultrasound is critical in these cases. Ultrasound allows accurate placement, helps avoid nerve injury by preventing a too lateral approach, can help avoid issues with plaque disruption and dissection by identifying less calcified areas of the artery, and subsequently, we believe, reduces the risk of delayed bleed or pseudoaneurysm formation. These issues are of particular concern when accessing the popliteal artery.
- When accessing the PA, the patient is usually placed prone (and thus should be able to tolerate this), and after access is achieved with a wire, the patient is repositioned supine for the CFA access and subsequent interventions. An open exposure of the CFA can also be utilized.
- Cross over can be difficult in patients with a sharp aortic bifurcation. Having achieved cross over, usually with a SOS Omni or Rim catheter (although a pigtail can be used), it is critical to remember to get a sheath that is stiff enough across the bifurcation (usually on a stiff wire) to maintain access.
- Finally, when the critical lesions are relatively distal in the SFA or involve below knee run-off, antegrade (going distal) ipsilateral approach helps reduce problems with wire manipulation etc. However, this can be quite difficult. A large pannus may prevent the access needle being able to be placed relatively “flat." This is important because the tendency of the wire is to go posterior and into the PFA. If this does occur, using a glide wire and directional catheter can allow re-manipulation into the SFA. In some settings, where it is desired to perform simultaneous iliac and distal SFA interventions, it is possible to use on access. In this technique, the CFA is access essentially at right angles, and the wire passed distally. After the proposed intervention is performed, using a SOS Omni catheter, the sheath is pulled back until the SOS engages the edge of the access site. A wire is then advanced proximally, and the catheter re-directed and subsequently the sheath. The disadvantage of this approach is that the right-angled access can make interventions more difficult.
Anesthesia and Anti-coagulation
- In the majority of cases local anesthesia and intravenous sedation is sufficient. If the patient requires a prone approach and can tolerate that, one should consider general anesthesia or alternative strategies.
- Heparinization should be instituted at the time any sheath is in place across the aortic bifurcation or any catheter or sheath that is occlusive. In general, we aim for activated clotting times of 200-250 seconds.
Closure
- Clearly, in the majority of cases, closure has been achieved by either direct pressure or, in the case of open exposure, direct repair of the artery.
- Direct compression requires dedicated attention to making sure the coagulation parameters are normal, that blood pressure is controlled, and that direct pressure can be brought to the specific area of the CFA accessed. Patients who are obese and/or hypertensive present special problems, as do those who cannot lie still for the request 4-6 hours. The most important aspect is that whoever is holding compression is really over the artery puncture site. This can be done simply with two or three fingers, but it is not uncommon to find the person to whom the responsibility of holding pressure has been given, of blindly forcing a fist over the general groin area resulting not only in inadequate compression, but increased pain, etc.
- A number of closure devices are available that, depending on the quality of the artery and size of sheath used for intervention, can permit closure and allow more rapid mobilization of the patient. Critical to these uses are confirmation that the CFA is accessed and that the site is not too heavily calcified. If it is recognized that the artery is calcified, it is often safer to plan an open exposure. The STAR® (Abbott Labs, Redwood, CA) closure can close sheaths of 6 and occasionally 7 Fr diameter, the Proglide® (Abbott Labs, Redwood, Ca) 6-8Fr., and the Prostar® 8-10 Fr. diameter access sites. A variety of devices have been utilized for percutaneous closures with good results. These devices are designed to place intra-arterial sutures that are then tied down at the end of the case [11, 12]. Many clinicians prefer to close over a glide wire at the end so that if there is a failure of closure, a sheath can be re-introduced and a cut down performed. This is more often an issue when aortic or iliac lesions are being addressed due to the larger size of the sheaths and devices used.
Techniques
Angioplasty:
Angioplasty is based on the concept of creating a controlled disruption of plaque while simultaneously stretching out the medial and adventitial layers of the artery. Optimal results are associated with short, focal lesions. Circumferential calcium is associated with an increased risk of rupture and ostial and eccentric lesions have higher failure rates.
- Angioplasty in the femoral popliteal vessels has been associated with patency rates of 65-77% at 1 year, 42-55% at 5 years depending of TASC lesion status [13, 14]. Outcome is affected by run-off. In the STAR registry lesions with 3-vessel run off were associated with a 53% success rate 5 years post angioplasty vs. 16% if there was limited run-off. If the presentation is one of critical limb ischemia, the success rates decrease further, with 1 year success rates of 40% reported [15]. However, as a limb salvage technique it still may have great value, with immediate technical success rates as high as 88%.
- Currently, we use angioplasty in the setting of small distal below knee lesions or as a temporizing measure in critical limb ischemia when we do not feel other techniques are safe. Finally, it can be appropriate in the management of in-stent stenosis. Overall, we do not usually use angioplasty alone as the primary treatment because we feel the recurrence rate in most instances are increased compared to other techniques, particular with longer lesions [1].
Subintimal Angioplasty:
- Crossing total occlusions can be difficult. A number of options are available. The Frontrunner® (Cordis) or Quickcross® catheters are designed to maintain the wire in the center of the lumen and penetrate the plaque and/or thrombus in a controlled fashion. However, the most common technique, which may actually result in better patency, is to create a subintimal dissection plane [16-18].
- We most commonly use a glide wire, and by buckling the wire the subintimal plane is entered. Following with an angled glide catheter, the goal is to re-enter the lumen distal to the obstruction. This latter step is often the limiting factor. There are adjuncts to assist this, such as the Outback® or Pioneer® catheter which allow an angled needle to puncture back into the true lumen. Angiography confirms position, and a wire is advanced through the needle.
- Other adjuncts, such as catheters with endovascular probes are also being trialed for the management of chronic total occlusions. Although there are variations in results, 12-month patency rates are reported as high as 73% and limb salvage 94% including patients with tissue loss [19-21]. As with other interventions, increasing length of occlusion and reduced run off vessels diminish the success rates [19]. Having crossed the lesion stenting can be performed as well.
Cryoplasty:
- Cryoplasty is based on the premise that combining cold thermal energy with angioplasty will promote medial “stiffness” and plaque micro-rupture leading to a more uniform a smooth dilation with reduced risk of dissection. In addition, induction of smooth cell apoptosis may reduce recurrent hyperplasia [1]. Fava and colleagues reported on 15 patients who had lesions that were on average 86% stenotic and 6.5 cm long with runoff vessel patency averaging 1.7 patent trifurcation vessels. At 14 months there was 83% patency [22]. Laird, et al., studied 102 patients with SFA lesions, excluding those with critical limb ischemia and/or heavily calcified vessels. Initial success was 86% and at 9 months clinical success was 83% [23, 24].
- We have used the Polar Cath® (Boston Scientific, Natick, MA) as a stand alone technique. It can be used through sheaths that are slightly smaller than required to place stents of the same size, can be used in ostial lesions, and is useful in tibioperoneal vessels.
Bare Metal Stents:
- Nitinol stents have been associated with improved results compared to stainless-steel stents possibly due to greater radial force and reduced shortening when deployed [25].
- Nitinol stents have been associated with improved results compared to angioplasty alone in the SFA. In one trial, one year restenosis with angioplasty was 63% vs. 37% with nitinol stenting [25]. The majority of stents used in the peripheral vascular tree are self-expanding rather than balloon-fixed delivery systems. These stents can shorten but are more flexible. The SIROCCO trial documented a 24 month restenosis rate of 21% for self-expanding stents in femoral-popliteal arteries (with preservation of ABI) [26, 27].
- Because late restenosis secondary to intimal hyperplasia remains an issue, there has been interest in drug-eluting stents. To date, there has not been convincing data that drug-eluting stents in the femoral-popliteal arteries have greater patency rates [26].
Polytetrafluoroethylene (EPTFE)-covered stents:
- In an attempt to manage neo intimal hyperplasia, EPTFE stents have been studied. These presumably will prevent in growth through the stent mesh, but can cover critical branch vessels [1].
- The Viabahn® (AWL Gore & Associates, Flagstaff, AZ) is one such EPTFE-covered self expandable stent. Primary patency rates in the femoral-popliteal arteries at 2 years have been reported to be 74% [28].
- Eudora, et al., performed a prospective randomized study comparing these stents to above knee bypass with prosthetic material [29]. The covered stents had the same patency at one year, with significantly reduced length of stay (0.9 days vs. 3.1 days).
- Covered stent that come up to the origin of the superficial femoral artery may impede flow into the profunda. This area can be managed by angioplasty, cytoplast, treatment with an uncovered stent or surgical angioplasty.
Atherectomy:
- The Silver Hawk® (Fox Hollow, Redwood City, CA) allows directional mechanical de bulking of lesions. The TALON registry describes results among a complex population, with 1/3 suffering critical limb ischemia and 1/2 being diabetic. Freedom from secondary revascularization at one year was reported to be 80% [30]. Further follow up studies suggested a primary patency rate of 60% at 2 years in a variety of infra-inguinal interventions with 80% secondary patency [31].
- Of note, between 25-50% of cases will require adjunctive angioplasty or stenting at the same time [30-33].
- Thereto can be difficult, and associated with an increased risk of embolization or dissection, if the target lesion is heavily calcified.
- Our bias is that thereto is best suited for short lesions, although at times we have used it for longer lesions prior to stenting to either allow enough of a channel to cross the obstruction with the stent or if we feel that de bulking will prevent compression of the stent.
Laser Atherectomy:
- Lasers which use photochemical energy (thus reducing the risk of damage to adjoining tissue) appear to be ideally suited for long, relatively concentric lesions [1, 34].
- The TURBO Elite® (Spectrometric, Colorado Springs, CO) is currently available, but the largest diameter is 2.5 mm, and in the majority of cases adjunctive measures are needed (either rebellion and/or stenting).
- Lasers appear particularly suited patients with critical limb ischemia at high surgical risk, although nearly 1/2 will require stenting at the time of the procedure [34, 35].
Post intervention management:
- Patients should be maintained on E CASA immediately post-procedure.
- Stents or lumens <7 mm should further be managed by the addition of clopidogrel for 6-12 months at least.
Pitfalls and Complications
Dissection
- Dissection can occur during angioplasty or if a wire enters a subintimal plane.
- Small "distal pointing" flaps can be observed but those with a proximal edge or if there is any suggestion of rupture and/or luminal compromise, should be treated.
- Initial treatment can be simple angioplasty, but in the majority of cases stenting appears to more reliably “take down” the flap and reduce the risk of complications [25].
Embolism
- Embolism occurs predominantly in two settings: crossing and opening an occluded artery in which there has been buildup of thrombus and/or in debulking a heavily calcified lesion. This significance of distal embolization is increased depending on the quality of the below knee run-off.
- Patients with single vessel run-off can experience immediate threat to limb viability. Soft micro emboli can result in extensive difficult to treat digital occlusion resulting in chronic pain and threatening tissue viability.
- When attempting to cross an occluded vessel in which the clinical setting suggests that there may be chronic thrombus, it is advisable to consider a trial of lytic therapy prior to definitive intervention. Ideally, a perfusion catheter can be advanced across the area of concern. Failing that, "parking" a catheter just proximal to the area of occlusion can suffice.
- If there is concern about distal embolization at the time of intervention, a distal protection device can be advanced prior to intervention. This is particularly of use when performing atherectomy of a relatively calcified lesion.
- Finally, if distal embolization does occur, options include lysis, trying to use suction embolectomy or mechanical lysis [28, 36].
Psuedoaneurysm
- CFA psuedoaneurysm occurs in patients who are hypertensive and/or in severely calcified vessels.
- Usually the lesion is detected acutely because of concern of bruising or persistent oozing from the access site but they can present in a delayed fashion as a “knot” or pulsatile mass in the groin.
- Initial assessment is by ultrasound.
- Psuedoaneuryms, in particular acute ones, are more often than not management by ultrasound guided thrombin injection. The requirement for this is that the neck be narrow and easily seen. Usually less than 0.3 cc is required. At the end of the injection, repeat duplex distally is performed to rule out embolism. Very small lesions can be observed.
- Larger mature lesions may require surgical repair because of the risk of creating a nidus for subsequent embolic complications.
Stent-Fracture:
- Stent fracture has been recognized as a major limitation in treating lesions that cross the hip or knee joints. Even if this is not the case, stent fracture has been documented in up to 1/4 of patients (at 10 month follow-up) with 1/3 of these resulting in occlusion, and a further 1/3 resulting in significant stenosis [37]. Fracture is more common the longer the lesion treated, and reduces 1-year primary patency by 1/2 [37].
- Treatment includes re-stenting or bypass.
Overview of the Procedure
- In most cases access is obtained using a 21 gauge needle and 0.18 wire (4F micropuncture set).
- Ultrasound can greatly assist in directly accessing the least diseased portion of the vessel.
- A 4 or 5-Fr sheath is advanced.
- If performing a cross over access, having achieved this, remember to (a) heparinize prior to placing the working sheath across the bifurcation and (b) use a stiff wire while placing the sheath to prevent collapse of the sheath if the bifurcation is very angulated.
- If performing an antegrade approach, perform an angiogram once the micropuncture sheath is place to confirm location and anatomy.
- If there is significant risk of embolization and there is only one vessel runoff, consider using a distal embolic protection device.
- Use road map or smart mask imaging technique if having problem crossing the target vessel.
- If a ruler or anatomy does not help clearly identify the area to be treated (e.g., prior to stent deployment), use road map or smart mask imaging technique.
- Always perform a completion angiogram, and if extensive disease, this should include the entire limb.
Nuts and Bolts
- These are by no means all of the devices available but they are a good representation.
- GORE VIABAHN ENDOPROSTHESIS STENT
The GORE VIABAHN Endoprosthesis is a flexible self-expanding PTFE lined nitinol stent.
Device Sizing Sheath Size (fr)
| Device Diameter (mm) |
Lumen |
2.5 cm |
5 cm |
10 cm |
15 cm |
Guidewire |
Balloon |
|
5 |
4.0 - 4.7 |
8 |
8 |
8 |
8 |
.035 |
5.0 |
|
6 |
4.8 – 5.5 |
8 |
8 |
8 |
8 |
.035 |
6.0 |
|
7 |
5.6 – 6.5 |
9 |
9 |
9 |
9 |
.035 |
7.0 |
|
8 |
6.6 – 7.5 |
9 |
9 |
9 |
9 |
.035 |
8.0 |
|
9 |
7.6 – 8.5 |
11 |
11 |
11 |
11 |
.035 |
9.0 |
- CORDIS S.M.A.R.T. CONTROL STENT
The Cordis S.M.A.R.T. CONTROL Nitinol Stent system is designed to deliver a self-expanding stent via a 6Fr sheathed delivery system. The self-expanding stent is composed of a nickel titanium alloy (nitinol). A total of 12 (6 at each end) tantalum radiopaque markers are located on the ends of the stent. The stent is a flexible, fine mesh tubular prosthesis, which achieves its unconstrained diameter upon deployment. Upon deployment, the stent imparts an outward radial force on the luminal surface of the vessel to establish patency.
Stent Size Selection Table
Vessel Lumen Diameter Unconstrained Stent Diameter % Foreshortening
4.0 – 5.0 mm 6 mm 1.1 %
5.0 – 6.0 mm 7 mm 1.8 %
6.0 – 7.0 mm 8 mm 2.8 %
7.0 – 8.0 mm 9 mm 4.0 %
8.0 – 9.0 mm 10 mm 5.5 %
- Spectranectics TURBO Elite Laser Catheter
When laser is ready to be used:
- Plug in catheter and calibrate.
- Connect Toughey and Clicker and attach to pressure bag.
- Set Fluence and Rate (see chart below).
- Insert catheter over .014 wire (eg: Mailman) using the .014 wire allows saline to flow easier.
- When ready to laser, ensure saline is flowing and press the Ready button.
- Move laser catheter ½ to 1mm per second. NO FASTER.
- Size the catheter to 2/3 the size of the vessel.
Catheter Size Start 2nd,3rd,4th Calcium Saline
Passes Protocol
Fluence/Rate Fluence/Rate Fluence/Rate
.09mm(5FR) 50/40 60/60 80/80 20 cc syringe
1.4mm(5FR) 50/40 60/60 60/80 20 cc syringe
1.7mm(5FR) 50/40 60/60 60/80 20 cc syringe
2.0mm(7FR) 50/40 60/60 60/80 Pressure bag
2.3mm(7FR) 50/40 60/60 60/80 Pressure bag
2.5mm(8FR) 45/50 45/65 45/80 Pressure bag
Turbo Booster 45/45 45/45 45/80 Pressure bag
Remember:
7FR Turbo Booster works with 1.4 mm to 1.7 mm catheters
8FR Turbo Booster works with 1.4 mm to 2.0 mm catheters
- POLARCATH PERIPHERAL DILITAION SYSTEM
Balloon Diameters:
0.035" – 4 thru 8mm
0.014" – 2.0mm, 2.5mm, 3mm, 3.5mm, 4mm, 5 mm, 6 mm
Balloon Lengths:
0.035" – 20, 40, 60, 80 & 100mm
0.014" – 20, 40, 60 & 100mm
Catheter Lengths:
0.035" – 80cm & 120cm
0.014" – 135cm & 150cm
Non-Expanded Profile:
0.035" – 0.078" (7F)
0.014" – 0.055" (5.8F)
Sheath Compatibility
0.035" - 4mm and 5mm - 6F
6mm and 7mm - 7F
8mm - 8F
0.014" - 2mm thru 4mm - 5F
5mm & 6mm - 6F
Example Cases
 |
|
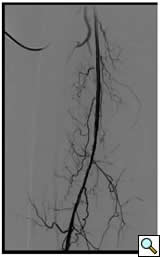
Figure 1A: Angiogram |
 |
|
Figure 1B: Following a day |
 |
| Figure 1C: Angiogram shows restoration of limb perfusion. |
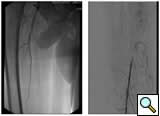
Case 1: An 84-year-old male with rest pain, who had undergone prior revascularization with covered stents, represents with recurrent rest pain. The prior stents had come close to the origin of the SFA and at that time there had been some narrowing. Exposure was provided by performing a cut down and patching the CFA-proximal SFA to the level of the stents, following which gentle thrombectomy was performed, and a wire passed down. The angiogram demonstrated a distal lesion at the adductor canal which contributed to the thrombosis (Figure 1A). The patient underwent a day of lysis, following which cryoablation was used to treat the distal lesion (Figure 1B) and completion angiogram showed restoration of limb perfusion (Figure 1C).
Case 2: A 74-year-old male with new onset severe claudication and threatened toes on the right side. An up-and-over wire was able to be advanced into the superficial femoral artery (but was totally occlusive). Cryoablation restored patency (Figure 2).
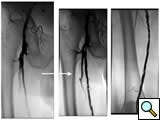 |
| Figure 2 |
Case 3: An 81-year-old male with rest pain. The patient underwent angiography via an antegrade approach, which revealed extensive
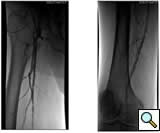 |
| Figure 3A |
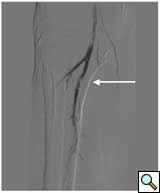 |
| Figure 3B |
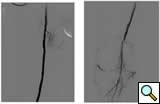 |
| Figure 3C |
calcific lesions of the superficial femoral artery (Figure 3A) and a lesion in the tibio-peroneal trunk (Figure 3B). The SFA lesions were debulked with a laser and the patency restored with Viabahn stents (Figure 3C). The tibio-peroneal represented the sole run off and was left as we were worried about creating a disaster if the lesion ruptured or dissected. ABI increase from 0.19 pre-procedure to 0.55 post and the patient is now able to ambulate with manageable claudication. Options to treat the more distal lesion could involve using a distal protection device and coronary balloons and/or stents.
Case 4: A 67-year-old woman with diabetes and tissue loss in the foot secondary to pressure injury with poor healing. Angiogram through an antegrade approach shows a small vessel (average diameter 3 mm) with proximal occlusion and segmental stensoses (Figure 4A). After angioplasty followed by cryoablation, the proximal stenotic areas were treated with 5 mm diameter coronary self-expanding bare stents (Figure 4B). The foot has now healed.
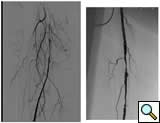 |
 |
| Figure 4A | Figure 4B |
References
- Perera GB, Lyden SP. Current trends in lower extremity revascularization. Surg Clin North Am 2007;87:1135-47.
- Aquino R, Johnnides C, Makaroun M, et al. Natural history of claudication: long-term serial follow-up study of 1244 claudicants. J Vasc Surg 2001;34:962-70.
- Muluk SC, Muluk VS, Kelley ME, et al. Outcome events in patients with claudication: a 15-year study in 2777 patients. J Vasc Surg 2001;33:251-57.
- Tendera M, Wojakowski W. Role of antiplatelet drugs in the prevention of cardiovascular events. Thromb Res 2003;110:355-59.
- Agnelli G. Rationale for the use of platelet aggregation inhibitors in PAD patients. Vasc Med 2001;6:13-15.
- CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996;348:1329-39.
- Harker LA, Boissel JP, Pilgrim AJ, Gent M. Comparative safety and tolerability of clopidogrel and aspirin: results from CAPRIE. CAPRIE Steering Committee and Investigators. Clopidogrel versus aspirin in patients at risk of ischaemic events. Drug Saf 1999;21:325-35.
- Easton JD. Evidence with antiplatelet therapy and ADP-receptor antagonists. Cerebrovasc Dis 2003;16:I20-6.
- Rutherford RB. Standards for evaluating results of interventional therapy for peripheral vascular disease. Circulation 1991;83:I6-11.
- Becker F. Exploration of arterial function with noninvasive technics. Results in chronic arterial occlusive disease of the lower limbs according to Leriche and Fontaine classification. Int Angiol 1985;4:311-22.
- Tehrani HY, Peterson BG, Katariya K, et al. Endovascular repair of thoracic aortic tears. Ann Thorac Surg 2006;82:873-78.
- Morasch MD, Kibbe MR, Evans ME, et al. Percutaneous repair of abdominal aortic aneurysm. J Vasc Surg 2004;40:12-16.
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease. Int Angiol 2007;26:81-157.
- Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg 2007;45:S5-67.
- Clark TW, Groffsky JL, Soulen MC. Predictors of long-term patency after femoropopliteal angioplasty: results from the STAR registry. J Vasc Interv Radiol 2001;12:923-33.
- Nadal LL, Cynamon J, Lipsitz EC, Bolia A. Subintimal angioplasty for chronic arterial occlusions. Tech Vasc Interv Radiol 2004;7:16-22.
- Bolia A, Sayers RD, Thompson MM, Bell PR. Subintimal and intraluminal recanalisation of occluded crural arteries by percutaneous balloon angioplasty. Eur J Vasc Surg 1994;8:214-19.
- Bolia A, Brennan J, Bell PR. Recanalisation of femoro-popliteal occlusions: improving success rate by subintimal recanalisation. Clin Radiol 1989;40:325.
- Lazaris AM, Salas C, Tsiamis AC, et al. Factors affecting patency of subintimal infrainguinal angioplasty in patients with critical lower limb ischemia. Eur J Vasc Endovasc Surg 2006;32:668-74.
- Lipsitz EC, Ohki T, Veith FJ, et al. Does subintimal angioplasty have a role in the treatment of severe lower extremity ischemia? J Vasc Surg 2003;37:386-91.
- Lipsitz EC, Veith FJ, Ohki T. Subintimal angioplasty in the management of critical lower-extremity ischemia: value in limb salvage. Perspect Vasc Surg Endovasc Ther 2005;17:11-20.
- Fava M, Loyola S, Polydorou A, Papapavlou P, Mendiz O, Joye JD. Cryoplasty for femoropopliteal arterial disease: late angiographic results of initial human experience. J Vasc Interv Radiol 2004;15:1239-43.
- Laird JR, Biamino G, McNamara T, et al. Cryoplasty for the treatment of femoropopliteal arterial disease: extended follow-up results. J Endovasc Ther 2006;13:II52-59.
- Laird J, Jaff MR, Biamino G, et al. Cryoplasty for the treatment of femoropopliteal arterial disease: results of a prospective, multicenter registry. J Vasc Interv Radiol 2005;16:1067-73.
- Schillinger M, Sabeti S, Loewe C, et al. Balloon angioplasty versus implantation of nitinol stents in the superficial femoral artery. N Engl J Med 2006;354:1879-88.
- Duda SH, Bosiers M, Lammer J, et al. Drug-eluting and bare nitinol stents for the treatment of atherosclerotic lesions in the superficial femoral artery: long-term results from the SIROCCO trial. J Endovasc Ther 2006;13:701-10.
- Duda SH, Bosiers M, Lammer J, et al. Sirolimus-eluting versus bare nitinol stent for obstructive superficial femoral artery disease: the SIROCCO II trial. J Vasc Interv Radiol 2005;16:331-38.
- Jahnke T, Andresen R, Muller-Hulsbeck S, et al. Hemobahn stent-grafts for treatment of femoropopliteal arterial obstructions: midterm results of a prospective trial. J Vasc Interv Radiol 2003;14:41-51.
- Kedora J, Hohmann S, Garrett W, Munschaur C, Theune B, Gable D. Randomized comparison of percutaneous Viabahn stent grafts vs prosthetic femoral-popliteal bypass in the treatment of superficial femoral arterial occlusive disease. J Vasc Surg 2007;45:10-16.
- Ramaiah V, Gammon R, Kiesz S, et al. Midterm outcomes from the TALON Registry: treating peripherals with SilverHawk: outcomes collection. J Endovasc Ther 2006;13:592-602.
- Zeller T, Sixt S, Schwarzwalder U, et al. Two-year results after directional atherectomy of infrapopliteal arteries with the SilverHawk device. J Endovasc Ther 2007;14:232-40.
- Zeller T, Rastan A, Schwarzwalder U, et al. Midterm results after atherectomy-assisted angioplasty of below-knee arteries with use of the Silverhawk device. J Vasc Interv Radiol 2004;15:1391-97.
- Zeller T, Rastan A, Schwarzwalder U, et al. Percutaneous peripheral atherectomy of femoropopliteal stenoses using a new-generation device: six-month results from a single-center experience. J Endovasc Ther 2004;11:676-85.
- Scheinert D, Laird, Jr JR, Schroder M, Steinkamp H, Balzer JO, Biamino G. Excimer laser-assisted recanalization of long, chronic superficial femoral artery occlusions. J Endovasc Ther 2001;8:156-66.




