ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Esophagectomy with Three-Field Lymph Node Dissection
Patient Selection
Three-field dissection (TFD) for squamous cell carcinoma of the esophagus has been practiced by Japanese surgeons since the early 1980's. Several reports, exclusively from Japan, showed that approximately one third of patients had previously unsuspected metastases to the cervical nodes. In 1997, we initiated a small pilot trial to evaluate the feasibility of the technique and to assess the prevalence of nodal metastases to the cervical lymph nodes in our patient population. In a preliminary report, we observed that occult metastases to the cervical nodes occurred in 30% of patients regardless of cell type. The procedure has since become our approach of choice for nearly all patients with esophageal cancer.
Esophagectomy with TFD is the procedure of choice at our institution for nearly all esophageal cancer patients provided the surgeon believes that a complete resection of all gross disease can be accomplished. Thus, patients with visceral metastases (liver, lung, etc.) detected prior to or during operations are not eligible. Similarly, patients with clearly documented T4 disease (detected preoperatively or intraoperatively) are not considered candidates for TFD. The presence of extensive nodal disease is not considered a contraindication to resection unless it clearly and unequivocally extends beyond the proposed fields of dissection. Although some surgeons have advocated a possible benefit of TFD in patients with palpable cervical nodes, we have no experience in this regard.
We have limited the use of TFD to tumors located within the tubular esophagus regardless of cell type. Tumors of the lower third are considered only if they do not extend past the gastroesophageal junction. Patients with tumors located precisely at the gastroesophageal junction are not considered for TFD at the present time.
Given the magnitude of the dissection and the potential for significant perioperative fluid shifts, patients are carefully evaluated to determine their ability to withstand the planned operation. Generally, patients with an FEV1 less than 1.5 liters despite smoking cessation, aggressive physiotherapy, and bronchodilators, are not eligible for resection and are referred for non-operative therapy. Cardiac disease, if clinically suspected, is carefully assessed using non-invasive means or angiocardiography if necessary. Age by itself is not a major consideration, however we have not attempted the operation in otherwise good candidates over 75 years old.
Operative Steps
The procedure is carried out through 3 incisions: a right thoracotomy followed by a laparotomy and collar neck incision.
The Thorax
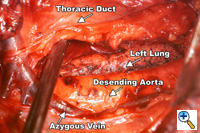 Figure 1. View from a right thoracotomy. Specimen is mobilized anteriorly along the descending thoracic aorta including the thoracic duct. Figure 1. View from a right thoracotomy. Specimen is mobilized anteriorly along the descending thoracic aorta including the thoracic duct. |
A right fifth interspace thoracotomy is performed regardless of the location of the tumor within the esophagus. The "first field" comprises the middle and lower mediastinum and is bounded superiorly by the tracheal bifurcation, inferiorly by the esophageal hiatus, anteriorly by the hilum of the lung and pericardium and posteriorly by the descending thoracic aorta and the spine. The en-bloc resection of the middle and lower mediastinum begins by incising the mediastinal pleura over the anterior aspect of the azygous vein from the level of the azygous arch superiorly to the aortic hiatus inferiorly. The dissection proceeds leftwards anterior to the aorta and across the mediastinum to the opposite pleura, which is entered along the entire length of the incision. The thoracic duct is thus mobilized anteriorly towards the specimen and is ligated inferiorly at the aortic hiatus and superiorly as it crosses to the left side of the mediastinum (Figure 1). The arch of the azygous vein, but not its main trunk, is resected en-bloc with the specimen. The anterior dissection is commenced by division of the azygous vein at its caval junction and by carrying the dissection along the right main bronchus and the posterior aspect of the hilum of the right lung. The hilar and subcarinal nodes are cleared and a patch of pericardium is resected en-bloc with the tumor-bearing esophagus if the latter abuts the pericardial sac.
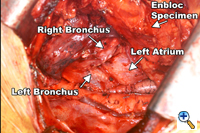
Figure 2. The en-bloc specimen is completely mobilized, revealing the left lung, the tracheal bifurcation and the pericardium. |
Division of both pulmonary ligaments (left and right) completes the esophageal mobilization (Figure 2). For tumors traversing the hiatus, a one-inch cuff of diaphragm is circumferentially excised en-bloc with the specimen using electrocautery. The completed dissection clears all nodal tissue in the middle and lower mediastinum including the right and left paraesophageal, parahiatal, para-aortic, subcarinal, bilateral hilar, and aortopulmonary lymph nodes.
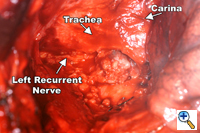
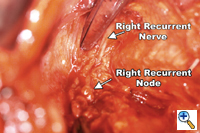
Dissection of the "third-field" begins during the thoracic portion of the procedure and is later completed through a collar neck incision. Dissection of the nodes in the superior mediastinum includes the nodes along the right and left recurrent laryngeal nerves throughout their mediastinal course. The paratracheal retrocaval compartment is not disturbed. The left recurrent nerve is dissected using a "no-touch" technique and nodes along its anterior aspect are carefully excised (Figure 3). Notably, there is a paucity of nodal tissue along the left nerve in nearly all Caucasians. The right recurrent nerve is carefully exposed near its origin at the base of the right subclavian artery. The right recurrent nodal chain begins at that level and forms a continuous package that extends through the thoracic inlet to the neck. Again, the nerve is dissected using a strict "no-touch" technique (Figure 4). Through the cervical incision, the remainder of the recurrent nodes are dissected as are the lower deep cervical nodes located posterior and lateral to the carotid sheath. Thus the "third-field" includes a continuous anatomically inseparable chain of nodes that extends from the superior mediastinum to the lower neck. These nodes should be appropriately labeled cervicothoracic (CT) nodes rather than cervical nodes.
The Abdomen
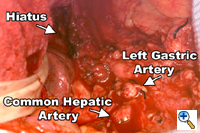 Figure 5. This view of the upper abdomen reveals the extent of the retroperitoneal lymphadenectomy superior to the upper border of the pancreas. Figure 5. This view of the upper abdomen reveals the extent of the retroperitoneal lymphadenectomy superior to the upper border of the pancreas. |
The abdomen is entered through a mid-line incision. The omentum is separated from the colon in the avascular plane and the lesser sac is entered. Following division of the short gastric vessels, the retroperitoneum is incised along the superior border of the pancreas. The retroperitoneal lymphatic and areolar tissues are swept superiorly towards the esophageal hiatus and medially along the splenic artery to the celiac trifurcation. The left gastric artery is divided flush with its celiac origin and the nodes along the common hepatic artery are dissected towards the specimen. This retroperitoneal dissection is bounded by the dissected esophageal hiatus superiorly, the hilum of the spleen laterally and the common hepatic artery and inferior vena cava medially (Figure 5). Finally, the lesser curvature and left gastric nodes are included with the specimen as the gastric tube is prepared. The omentum is resected as a separate specimen at least one inch outside the gastroepiploic arcade.
The Neck
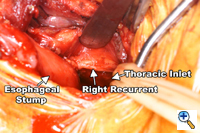
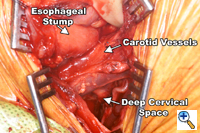
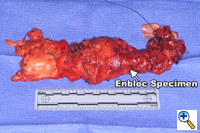
A generous low collar incision is performed and subplatysmal flaps are raised inferiorly and superiorly. The strap muscles and the medial heads of the sternocleidomastoid are divided. The esophagus (previously fully mobilized from the thorax) is retrieved from the prevertebral space. The esophagus is divided distally and the specimen is retrieved in the abdomen. The previously dissected recurrent nerves are easy to visualize (especially the right recurrent) and any residual nodal tissue is excised (Figure 6). Next, the nodes posterior and lateral to the carotid sheath are removed along with the supraclavicular nodes (Figure 7). The dissection is limited superiorly by the inferior belly of the omohyoid. Within the abdomen the gastric tube is prepared and the specimen is removed (Figure 8). Gastrointestinal continuity is restored by a cervical esophagogastrostomy.
Preference Card
- Standard thoracotomy and laparotomy trays.
- An upper hand retractor for abdominal exposure.
Tips & Pitfalls
- Clip or ligate all lymphatic channels between the thoracic duct and the spine to minimize the probability of a chylothorax.
- Never directly handle or encircle the recurrent nerves with a vessel loop for retraction purposes. A "no-touch" technique is essential to reduce the probability of recurrent nerve injury. In my experience, the incidence of recurrent nerve injury is 5-10%. No patient has required a tracheostomy for recurrent nerve injury.
- The right vagus nerve serves as a good guide to locate the right recurrent nerve. Attempts to directly locate the nerve are potentially hazardous.
- During the cervical portion of the operation, be particularly careful in identifying the recurrent nerves. Deprived of their supporting areolar tissues, the nerves tend to "wander" out of the tracheoesophageal groove.
- The pretracheal, retrocaval compartment should not be dissected. It is a rare site of nodal spread and its dissection deprives the trachea of an important source of collateral blood flow.
Hospital mortality is 5% and complications occur in 45% of patients. Occult metastases to the recurrent nerve nodes are seen in 37% of patients with adenocarcinoma and 34% of those with squamous cell cancer. Overall 5-year disease free survival is 46%.
References
- Altorki NK and Skinner D. Should en-bloc esophagectomy be the standard of care for esophageal carcinoma? Ann Surg 2001; 234:581-7.
- Altorki NK, Lerut T. Three-field lymph node dissection for cancer of the esophagus. Pearson FG, Cooper JD, Deslauriers J, Ginsberg RJ, Hiebert CA, Patterson GA, and Urschel HC Jr (eds): Esophageal Surgery, 2nd Edition. New York, Churchill Livingstone, 2002.
- Altorki NK, Kent M, Ferrara CA, Port JP. Three-field lymph node dissection for squamous cell and adenocarcinoma of the esophagus. Ann Surg 2002; 236:177-83.