Introduction
Safe performance of thoracic endovascular procedures including thoracic stentgrafting of aneurysms, dissections, retrograde delivery of transcatheter aortic valves, and other complex endovascular procedures for structural heart disease requires zero tolerance for major access-related complications. Thorough preoperative planning, understanding the pathology of aorto-iliac occlusive disease, advanced endovascular skills, and ability to perform an iliac conduit via a retroperitoneal approach are necessary to achieve such results. Furthermore, deliverability of complex thoracic endovascular devices through tortuous anatomy or old graft material may be improved by the more proximal access provided by an iliac conduit [1]. Details of the iliac conduit procedure will be described below as well as some additional tips to avoid access complications.
Indications and Preoperative Planning
Patient Selection
Currently, patients undergoing thoracic aortic endografting or transcatheter aortic valve procedures require access with devices ranging between 18 Fr to 30 Fr in caliber. Typically, the size-limiting segment is the external iliac arteries. Depending on the size of sheath required, a minimum diameter of 9 mm (3French ~ 1mm) may be necessary for safe access via the common femoral arteries. Although iliac anatomy can be assessed by angiography, these measurements are purely intraluminal and this area is best assessed with the use of computed tomography performed both with and without contrast to allow for true outer diameter measurements of the vessels and an understanding of the particular morphology of atherosclerotic disease. Good preoperative imaging not only provides information regarding the need for an iliac conduit but also details about iliac and aortic tortuosity that are important for site of delivery selection. Occasionally, the patient’s minimum iliac diameter is borderline acceptable and final determination about the need for a conduit can be made intraoperatively. First, common femoral access is obtained, then exchange for a stiff wire is made, and quality and elasticity of the vessels can be assessed by the sequential placement of large bore dilators (20 Fr to 24 French long taper hydrophilic dilators, Cook Inc., Bloomington, IN).
Operative Procedure
These procedures can be performed under regional anesthesia as well as general anesthesia. Radiolucent attachments for the fluoroscopy operating table are available to facilitate the use of the Omni retractor (Omni-tract Surgical, St. Paul, MN) and the selection of one side or the other is dependent upon the need for reconstruction/revascularization, quality of the iliac artery, history of previous surgery, etc.
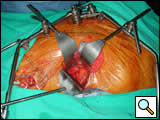
The incision is obliquely oriented approximately 1-2 fingerbreadths below the level of the umbilicus and lateral to the rectus abdominus (Figure 1). The external abdominal oblique is first divided in a muscle-sparing fashion in the direction of the fibers. The internal oblique muscle is likewise divided in the direction of the fibers, as is the transversus abdominus. Extra-peritoneal fascia and peritoneum are then retracted medially and dissection is carried out in the avascular plane of the retroperitoneum down to the level of the psoas muscle. All of the abdominal contents are then retracted medially with the help of the Omni retractor providing excellent exposure of the lower infrarenal aorta, common iliac artery, and iliac bifurcation (Figure 2). Great care is taken to both identify and avoid injury to the ureter.
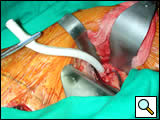
The vessels are then clamped both proximally and distally. This is often best achieved with the use of Satinsky-style clamps to apply anterior to posterior directed pressure on these vessels which often have significant posterior atherosclerotic plaque (Figure 3). Depending on the plan for completed reconstruction, the iliac vessel is either opened anteriorly for an end-to-side anastomosis or completely transected for an end-to-end anastomosis and a 10 mm by 30 cm graft is then anastomosed in typical fashion with running prolene sutures. Artery and graft are de-aired and the integrity of the anastomosis is confirmed. Then the graft is brought out of the incision and clamped (Figure 3).
Obtaining Access via the Conduit
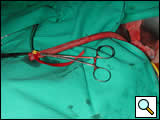
A 1cm nick is then made in the end of the graft to accommodate the side arm of the access sheath. A long 8 Fr sheath is then flushed and the suture tab is removed from the sheath port. The sheath is then advanced into the conduit with the side port coming through the previously made nick at the top of the graft, and an umbilical tape is tightened as a tourniquet around the hemostatic valve (Figure 4). Using wire and catheter exchange techniques, large bore delivery systems can then be delivered through the conduit in the usual fashion (Figure 5).
 |
 |
| Figure 4: Conduit access. | Figure 5: Device delivery via conduit. |
Once the procedure is completed, options include (1) tying off the conduit graft as a stump; (2) sewing the distal end of the conduit to the more distal iliac system in an end-to-end fashion as an interposition graft; (3) passing the distal end of the conduit under the inguinal ligament and performing either an end-to-end or end-to-side anastomosis to the common femoral artery. The last option is the best for patients who may require further intervention for diffuse thoracic aneurysmal disease as the conduit may be reused through a simple infrainguinal incision in the future.
Other Options
Using the same techniques described above, the conduit may be sewn in an end-to-side fashion to any portion of the infrarenal or thoracic aorta. Similarly, end-to-side grafting of a conduit to the axillary artery as described elsewhere to facilitate deep hypothermic circulatory arrest also provides excellent access to the thoracic aorta via the innominate [2].
References
- Abu-Ghaida AM, Clair DG, Greenberg RK, Srivastava S, O’Hara PJ, Ouriel K. Broadening the applicability of endovascular aneurysm repair: the use of iliac conduits. J Vasc Surg 2002;36:111-7.
- Sabik JF, Nemeh H, Lytle BW, et al. Cannulation of the axillary artery with a side graft reduces morbidity. Ann Thorac Surg 2004;77:1315-20.