ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Renal Artery Interventions
Tuesday, January 18, 2011
Patient Selection
- Renal artery stenosis (RAS) of >70% of one or both renal arteries AND
- poorly controlled hypertension despite multiple antihypertensive regimen, or
- chronic renal insufficiency related to RAS
- Dialysis dependent renal failure in patients without another definite cause
- Recurrent congestive heart failure or flash pulmonary edema not attributed to active ischemia
- Renal artery aneurysm (RAA) with a diameter greater than 2 cm, or aneurysmal renal artery with documented distal embolization and renal ischemia
- RAAs at risk for rupture in females contemplating pregnancy
- Acute dissection of the RA with malperfusion of RA
Operative Steps
- The patient is placed in the supine position. Both groins are prepped and draped. The procedure most commonly is performed under regional or local anesthesia.
- In majority of cases, common femoral artery is approached percutaneously or via femoral artery cutdown (retrograde approach). The brachial/radial artery (antegrade approach) is accessed only if there is difficulty in approaching from either of the femoral arteries, to avoid infra-renal abdominal aortic aneurysm, severe aortoiliac occlusive disease or severe caudal renal artery angulations.
- The 0.018-inch starter guidewire is advanced under fluoroscopic guidance into the aorta above the renal ostium (T12/L1). Renal arteries usually arise at top of L1 vertebral body.
- Once access (6 F sheath) is achieved, the patient is heparinized for a target activated clotting time ≥ 200 sec. This level of anticoagulation is maintained throughout the procedure.
- Start with an aortogram at L1-L2 using an Omniflush or Pigtail catheter with 10-15 ml of contrast. Renal arteries often originate either anteriorly of posteriorly, and initial aortogram may be improved with oblique views (20 deg LAO for right renal artery and vice versa).
- The artery is approached with the guide catheter (renal standard curve, IMA, renal double curve, hockey stick, SOS Omni, etc) in two ways: ”direct engagement” or “telescoping or no-touch”. In the ”direct engagement technique” the guide catheter is gently manipulated until it is seated in the renal artery. It is associated with higher rates of guide catheter induced dissection, embolization in ostial lesions. To minimize them, a “no-touch” technique maybe employed, wherein the guidewire is deployed in the abdominal aorta. Then the catheter is manipulated towards the renal artery ostium, and when the catheter is close to the ostium, the wire is removed allowing direct engagement of the guide catheter into the renal artery ostium.
-
In the “telescoping technique”, a 5 F diagnostic catheter is telescoped through a shorter 6-Fr guide catheter. The two catheters are advanced over the .018-inch guidewire and the diagnostic catheter is used to engage the ostium of the renal artery, once a wire has been placed across the stenosis, the diagnostic catheter is as a rail to introduce the guide catheter into the renal artery, and the diagnostic catheter is retracted thereafter. This minimizes the guide-catheter manipulations within the aorta and the renal artery ostium.
- Using an angled .018” Glidewire may be indicated to cross the lesion, however for use of hydrophilic wires in renal artery stenting the rule applies: “last in and first out”. Authors recommend switching back to stiffer .018-inch wire to reduce perforation risk.
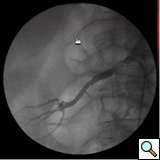
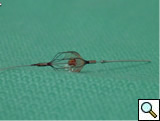
- Once the selective catheter is placed in the renal artery ostium, a selective arteriography may be performed (Figure 1), and translesional pressure gradients can be measured (operator choice). Nitroglycerine may be required to reduce renal artery spasm. An embolic protection device may be used at this point (operator choice, Figure 2).
- If the stenting is being done on a RAS, a small diameter (downsized) 4-6 mm compliant angioplasty balloon is inflated at the most proximal part of the renal artery stenosis and the renal artery is “pre-dilated”. The fully expanded native vessel diameter or the IVUS image will guide to optimal stent sizing.
- A premounted balloon-expandable stent (such as Express SD) is introduced through the sheath. Prior to deployment of stent, confirm the location of the stent in AP and oblique views to avoid missing the renal ostium.
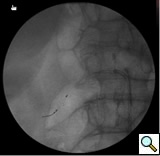
- Once the stent is deployed, a completion-angiogram is done to conform the proper placement of the stent and to make sure there has been no residual stenosis (Figure 3). It is important to make sure the renal ostium is properly covered by the stent or else it may undergo re-stenosis.
- After the completion angiogram, the introducer sheath is removed while carefully leaving the wire in the vessel. This wire is removed at last along with the introducer sheath. The femoral artery homeostasis is achieved by manual pressure or by using a closure device. Infrequently, the open exposed common femoral artery is repaired in standard fashion.
- The distal pulses are checked. If they correspond to preoperative baseline, the heparin is reversed. At this time, the sterile endovascular wires/catheters may be disposed.
Preference Card
Sheaths
- 5-6 F sheath, 10 cm long (Terumo Pinnacle Sheath) is used for femoral or brachial access.
- 5-6 F Ansel sheath, 45-55 cm (Cook Medical) may be used alternatively.
Catheters
- 5 F Bern (Boston Scientific 5F x 100cm Imager II Selective- Berenstein tip).
- 5 F pigtail catheter (Cook, Inc. Pigtail-Royal Flush Plus), 5 F x 90cm.
- Omniflush (Angiodynamics Angioptic), 4-5 F x 90 cm.
- 8.2 F IVUS catheter (Volcano, Vision PV), 8.2 F x 90 cm.
- 5-7 F x 65 cm hockey stick catheter, Internal mammary artery catheter, Multipurpose guiding catheter, Renal standard and double curve guiding catheter (Guidant Veripath)
Guidewires
- 0.018-inch x 200 cm TAD guidewire (Covidien)
- Bentson Starter guidewire (Boston Scientific 0.035-inch x 180 cm Starter).
- 180 cm hydrophilic guidewire J-curved (Terumo 0.018-inch x 180 cm Glidewire).
Balloons
- Angioplasty balloon (Boston Scientific Ultrathin Diamond Balloon), 2 cm length, 3.5-4 mm in diameter.
- Monorail balloons 3.5-4 mm diameter with 15-20 cm shaft
- Over the wire balloons 3.5-4 mm diameter with at least 80 cm shaft
- Smaller balloons with 1 mm or less diameter may be needed for initial dilatation.
Stents
- A large variety of balloon-expandable stents and stent grafts exists. This will be handled under “Nuts and bolts”!
Miscellaneous
- Luer-Lock (Boston Scientific FloSwitch HP).
- Needle for initial arterial access Micro-Puncture Introducer Set (Cook, Inc. 4 F, MPIS-401).
- Torque device ( Terumo Torque Device).
- Proglide Suture-Mediated Closure Device (Abbott Vascular, 5-8F access sites).
Tips & Pitfalls
- Documentation of the clinical indications and results of imaging and functional testing is a must.
- Periprocedural hydration, N-acetylcystein, and 2/3 or ½ contrast concentrations are important means to reduce contrast in patients with renal insufficiency.
- Balloon angioplasty is the treatment of choice for patients with fibromuscular dysplasia (10% of population), while stent is preferred in atherosclerotic renal artery stenosis (involves usually the proximal two cm of the renal artery).
- Stenting is recommended for atherosclerotic renal artery stenosis and dissection flaps in the renal artery. It is recommended to extend the stent 2 mm in the aorta and to flare the extending portion of the stent with a compliant balloon.
- Be aware of the position of the tip of the wire, and keep it at a fixed position for entire procedure. Arterial perforation or parenchymal injury is possible with bold wire advancements.
- Underdeployed stent is a common problem early in the operator’s experience. It is important to avoid postprocedural translesional pressure gradient. Intravascular ultrasound (IVUS) will visualize such cases and allow for better stent-intimal apposition. Furthermore, IVUS will reduce contrast use in patient with chronic renal insufficiency. Non-contrast IVUS-guided renal artery stenting is been practiced successfully in experienced hands. IVUS may guide the endovascular specialist in using embolic protection. Virtual histology based on IVUS images can detect significant atherosclerotic debris at risk for embolization. In these cases, embolic protection placed in distal renal artery will reduce embolic burden to the kidneys.
- Avoid bilateral renal artery stenting in one setting, if possible. A staged approach is preferred to reduce nephrotoxic effects and the contrast load.
- The skills necessary to perform renal interventions can not be attained through short “courses.” A dedicated formal training (six months minimum!) is necessary in order to obtain the “wire skills” and to independently perform these endovascular procedures. The trained endovascular surgeon should be able to deal with intraoperative complications of renal artery intervention including perforation, dissection and rupture. Peripheral artery angioplasty/stenting should be part of the armamentarium of the endovascular surgeon. The current privileging criteria in most tertiary hospitals require a minimum of 100 diagnostic, 50 interventional procedures to become certified/privileged in peripheral interventions.
- Most cardiothoracic surgeons will not meet the above training criteria (see also the credentialing section in the Endoluminal Technology Portal). In these cases, the necessary “wire skills” may be acquired by hiring a cardiothoracic surgeon who has had dedicated training in endovascular surgery. Alternatively, collaboration with other physicians with interventional skills (vascular surgeons, interventional radiologist/cardiologist) will be crucial.
- Train a dedicated nursing team for your endovascular cases. An Endosuite will substantially improve your imaging capabilities and broaden your endovascular options.
- All patients are placed on aspirin for 30-60 days unless there is a contraindication. Clopidogrel is not required for renal interventions, but may be used as alternative to aspirin.
- The patient will need to be followed up Duplex ultrasound in the postoperative period.
References
- Schneider PA. Endovascular Skills, 3rd Ed. New York: Informa 2009.
- Vascular Medicine and Endovascular Interventions. Rooke TW, et al ; Blackwell Futura;2007
- Mastery of Vascular and Endovascular Surgery. Zelenock GB, et al.; Philadelphia: Lippincott Williams and Wilkins; 2006.
- Peripheral Endovascular Interventions. White RA, et al.; New York: Springer; 1999.
- Endovascular Therapy: Principles of peripheral interventions. Lumsden AB, et al.; Malden: Blackwell Futura; 2006.
- Manual of Peripheral Vascular Intervention. Casserly IP, et al.; Philadelphia: Lippincott Williams and Wilkins; 2005.
- Vascular and endovascular surgery. Moore WS [Editor]; Philadelphia: Saunders; 2006.