Introduction
A vascular stent is a tube composed of a metal mesh intended to restore or preserve the patency of a vessel. The development of endovascular stent technologies made it possible and improved the minimally invasive treatment of many vascular diseases.
Indications for stent utilization include: dissection after angioplasty, residual stenosis (>30%), pressure gradient (>10 mmHg systolic), target vessel occlusion and recurrence of disease.
Each device implantation has its monetary value and the possibility for adverse events, and each intervention should be based on the solid clinical judgment of an endovascular specialist.
Complications of Stent Placement
Acute complications occur in the periprocedural period and include: arterial dissection, arterial rupture or occlusion, embolization of friable atherosclerotic material or stent migration.
Chronic complications of stent placement include recurrent stenosis (from intimal hyperplasia or disease progression in proximity to a stent), infection or stent damage from external forces.
Stent classification
Based on the method of deployment vascular stents are divided into two main categories: balloon expanded or self-expanding.
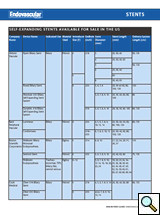 |
| Table 1: Balloon-Expandable Stents Available for Sale in the US. |
The balloon-expanded stents are straight, rigid cylinders constructed from stainless steel wire secured on an angioplasty balloon. Guiding catheters and sheaths are necessary to advance the device through tortuosity of the vascular system in order to avoid dislodgement of the undeployed stent. The prototypical Palmaz stent has undergone several modifications. Most balloon-expanded stents possess high radial force “hoop strength”, but lack flexibility. With improvement of mesh design-smaller cells and thinner struts flexibility has improved yet preserving excellent radial strength. The basic technique of stent deployment is as follows: Using wire guidance the appropriate sheath and dilator combination are passed through the lesion. After removing the dilator, the balloon catheter with the stent secured into place is advanced using fluoroscopy. Once in good position across the lesion the stent is expanded by inflating the balloon. The balloon first inflates at both ends of the stent followed by expansion in the center, allowing for precise positioning. Before deployment, it is critical to make sure that the stent is still correctly mounted on the balloon to avoid forward or backward misplacement. The working qualities of the balloon-expanded stent make it a better choice for precise treatment of a focal lesion in an area with less tortuosity and without repetitive external trauma such as the inguinal ligament and across joints. The excellent hoop strength makes it the preferred device for ostial lesions. As a routine the device size is matched to the anticipated vessel diameter. Balloon-expanded stents can be dilated 2-3 mm beyond their nominal size with appropriate balloon oversize. Corresponding decrease in the length should be expected.
Balloon-expanded stents are less flexible and despite the fact that the use of different size balloons is a possibility, optimal vessel wall apposition may not be achieved in an area of vessel size transition. Table 1 is a list of commercially available balloon-expandable stents in the U.S.
Self-expanding stents are usually constructed of stainless steel or the nickel-titanium alloy called nitinol. The former is not MRI compatible (only up to 1.5 tesla), while the later may be evaluated using MRI.
The Wallstent is composed of the thin Elgiloy stainless steel wire mesh, relying on predetermined spring like design to achieve desired expansion. Wallstents are compressed within a delivery catheter, which is an integral part of the delivery system. The external catheter maintains the collapsed state of the stent until its retraction allows for the device to expand. Device deployment is carried out by retracting the outer sheath while holding the stent in place with the inner tube. Due to its better flexibility Wallstents are preferred devices for tortuous vessels. They can cover longer lesions but are not practical for treating focal disease. These stents guarantee very precise placement only on the end to be deployed first (Wallstents deploy form distal to proximal). Definite advantage of the Wallstent is the ability to recapture the device until up to 85% of the stent is deployed. This may facilitate adequate repositioning. As a rule the leading end of the device is always maneuvered just past the planned landing zone, allowing for fine adjustments during its deployment. The final resting length of the Wallstent is difficult to predict in vivo.
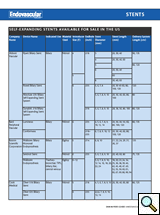 |
| Table 2: Self-Expanding Stents Available for Sale in the US. |
Nitinol stents are composed of a nickel-titanium alloy characterized by the property of thermal memory, result of the varying crystal lattice structure of the alloy at different temperatures. Nitinol stents exhibit minimal foreshortening, as much as 10%, and have a more predictable length once deployed in the body.
Self-expanding stents are better choices for tortuous vessels especially in areas of permanent external forces i.e. across the inguinal ligament or across joints etc. Because of the inability for additional dilatation of the device, as opposed to balloon-expanded stents, better device/target vessel size match is required.
In case of emergency self-expanding stents can be clamped with a shodden clamp. Any attempt to clamp a Palmaz stent will permanently deform the device.
Radiopacity is another feature of contemporary stents. Stainless steel devices are usually easy to identify. Nitinol stents on the other hand require radiopaque markers to facilitate accurate position and subsequent radiographic evaluation.
Most peripheral vessels can be stented through sheaths ranging from 6 to 10 French.
Table 2 is a list of commercially available self-expandable stents in the U.S.
Covered Stents
Fabric-covered stents are intended to replace the inner lining of a vessel by endovascular graft placement. There are several commercially available covered stents, with different approved indications. They usually require 8 to 12 Fr access sheaths.
| Fluency Plus | (Bard) | Tracheobronchial | Self-expanding |
| Jostent | (Abbott) | Coronary perforation | Balloon-expanded |
| Viabahn | (Gore) | SFA | Self-expanding |
| ICast | (Atrium) | Tracheobronchial | Balloon-expanded |
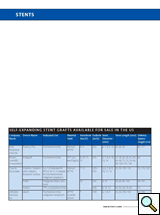 |
| Table 3: Self-Expanding Stent Grafts Available for Sale in the US. |
There are reports of off-label application of these devices for "salvage" femoral, popliteal or infrapopliteal interventions.
Drug-eluting stents are the latest advance in stent technology intended to counteract intimal hyperplasia and restenosis. The special coating provides controlled release of drugs blocking the activation of inflammatory and growth factors. Most data is derived from cardiac trials suggesting improved early and mid-term results, and several peripheral vascular studies are underway.
Table 3 is a list of commercially available covered stents (used for peripheral indications) in the U.S.
Aortic Endografts
The advances in the development of intravascular stents made possible the construction and approval of the currently available endovascular grafts for the treatment of abdominal and thoracic aortic pathology.
Currently, several second generation devices are approved for the treatment of abdominal aortic disease including ruptured AAA. Detailed characteristics and techniques of endovascular AAA repair are beyond the scope of this presentation.
Thoracic Endograft Devices are applicable in treating thoracic aortic pathology including aneurysms, traumatic aortic transection, complicated type B aortic dissection and also the different stages of the uncommon aortic wall abnormality of intramural hematoma, penetrating ulcer and pseudoaneurysm.
The Gore TAG Thoracic Endoprosthesis is currently the only FDA approved device for the treatment of thoracic aortic aneurysms in the US. The Pivotal Trial evidence demonstrated the advantages of lower operative mortality, less procedural blood loss, lower perioperative morbidity, shortened ICU and hospital stay and faster return to normal activity.
The GORE TAG endoprosthesis is comprised of an expanded polytetrafluoroethylene (ePTFE) tube reinforced with ePTFE/FEP (fluorinated ethylene propylene) film that is supported by a self-expanding nitinol exoskeleton. The endoprosthesis contains radiopaque gold marker bands sat the base of the device flares approximately 1 cm from each end of the endoprosthesis. An implantable ePTFE/FEP sleeve is used to constrain the endoprosthesis on the leading end of the delivery catheter. Unlacing of the endoprosthesis initiates in the middle of the device and simultaneously extends towards both ends of the endoprosthesis. The ePTFE/FEP sleeve remains between the exterior surface of the endoprosthesis and the intimal surface of the aorta. Both proximal and distal segments of the endoprosthesis have scalloped flares that improve the device/vessel wall apposition in tortuous segments of the thoracic aorta.
A variety of device diameters are available ranging from 26, 28, 31, 34, 37 and 40mm. There are three working device lengths: 10, 15 and 20 cm. Larger diameter sheaths (20-24 Fr) are available for device delivery.
There are several other thoracic endografts undergoing clinical trials for FDA approval:
The Talent Thoracic Device (Medtronic), The Valiant Thoracic Device (Medtronic), The Zenith TX2 TAA Endovascular Device, The Zenith TX2 Dissection Endovascular Device (Cook Medical), The Relay Thoracic Stent Graft (Bolton Medical).
Details of the technique of thoracic endovascular aortic repair (TEVAR) are discussed in a different section of the portal.
Summary
The minimally invasive vascular surgery approach has been facilitated by the development and constant improvement of the stent technologies. Broad variety of vascular disorders is currently treated with endovascular stents. The advances and improvements in the endovascular stents made it possible for the development and approval of the aortic endografts.
The endovascular specialist treating abdominal and thoracic aortic pathology has to be prepared to recognize and handle peripheral vascular complications and challenging situations. Acquiring and maintaining endovascular skills and being familiar with the different types of devices and their properties is a must for every specialist treating vascular diseases in order to provide optimal therapy.
References
- Rutherford RB. Rutherford Vascular Surgery, 6th ed. Philadelphia: Elsevier Saunders, 2005.
- Moore WS., Ahn SS. Endovascular Surgery,3rd ed. Philadelphia: W.B. Saunders, 2001.
- Schneider PA. Endovascular Skills, St. Louis: Quality Medical Publishing, 1998.
- Endovascular Today 2007, Volume 6, No 12 GORE TAG IFU, Flagstaff: W.L. Gore & Associates, Inc.




