ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Minimally Invasive Resection of Chest Wall Lymphangioma
Patient Selection
Lymphangiomas are benign lymph filled cystic lesions and may arise from lymphatics of the chest wall (1). These are typically asymptomatic unless they involve other structures—such as the thoracic outlet, which is a confined space of sufficiently small size to permit compressive symptoms. The axilla is a common site of occurrence. Most lymphangiomas present in childhood, and are thus managed by pediatric surgeons.
For patients who are symptomatic, surgical resection is the current mainstay of therapy, although sclerotherapy may have a role in more complex lesions or recurrences.
In general, minimally invasive techniques, when compared with their open counterparts, are associated with decreased time for recovery, shortened hospital stay, and less pain. Lymphangiomas, because of their underlying inherent properties—benign and fluid filled, make them ideal for a minimally invasive approach, in particular when involving the chest wall. However, there is limited experience with the use of such techniques in the management of these complex lesions.
Case
A 50-year-old male presented with a history of being evaluated for a right chest wall hematoma that occurred while he was lifting weights in the distant past. At that time, he described a bursting sensation during exercise and mild pain. He was evaluated at an outside emergency room, underwent cross sectional imaging, and was diagnosed with a chest wall hematoma.
Over the next eight years, he became progressively symptomatic from an enlarging mass encompassing his right chest wall and axilla. Magnetic resonance imaging revealed a cystic mass extending from the axilla along the chest wall involving the sub-pectoral space, between the pectoralis major and minor muscles. These findings were suspicious for a lymphangioma (Video 1).
Because of the inherent characteristics of lymphangiomas, the authors felt that a minimally invasive approach would be feasible. Given the paucity of these lesions, the reported experience using such an approach is limited. The ideal approach to these lesions is individualized based on each patient’s specific details.
Operative Steps
The authors thought that a minimally invasive approach would be feasible. However, this was inhibited due to the anatomic location of the cyst. Unlike the abdominal or thoracic cavities, which are amenable to the creation of a working space for a minimally invasive approach, the chest wall and its soft tissues do not contain a natural potential space. That being said, since lymphangiomas are typically fluid filled and easily decompressed, the authors felt that such a space could be created underneath the pectoralis muscle, utilizing decompression of this cystic lesion along with carbon dioxide insufflation (CO2).
Patient Positioning
The authors elected for a semi-supine position with the arm supported to open the axillary space (Figure 1A and 1B).
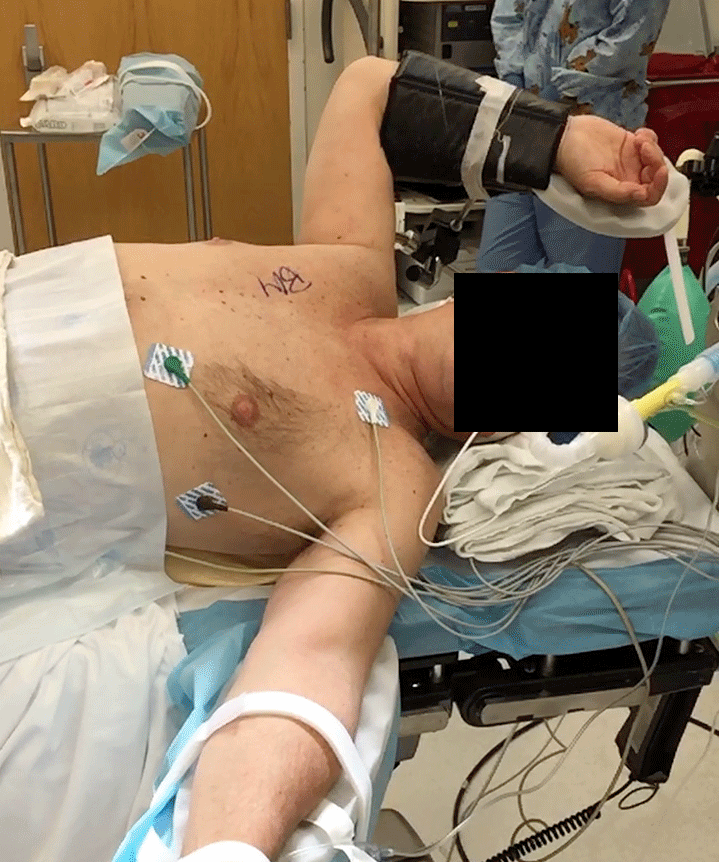
Figure 1A: Operative positioning; semi-supine position with the arm supported to open the axillary space.
Port placement
The authors' strategy for port placement was to select a peripheral area of the mass. To facilitate this, the most superficial area of the lesion was chosen. The authors placed a 5 mm balloon anchored trocar (Applied Medical, Rancho Santa Margarita, CA) under OPTIVIEW2 guidance to dissect an initial plane for insufflation with CO2 to obtain working room with decompression of the cyst. Initially, the port was inadvertently placed into the pleural space, this was recognized and the trajectory of the port altered to insure that the port abutted the wall of the lymphangioma (Video 2).
Operation
Once the wall of the lymphangioma was bluntly dissected under camera guidance, the cyst was entered with the port and the majority of its contents were aspirated. Insufflation was then used to create the working space for the dissection. The authors planned the trajectory of the ports and view to give optimal exposure of the neurovascular structures to avoid injury. Once a space was created, additional ports were placed along the perimeter of the dissection for the dissection. The borders of the lymphangioma were identified and dissected. The medial border was along the insertion of the pectoralis major muscle, and the superior border was the axillary vessels. The neurovascular bundle to the pectoralis major muscle was clearly observed coursing in a perpendicular fashion to the chest wall, and was protected throughout the dissection. The borders of the cyst were clarified during the procedure by insufflation of the lymphangioma (Video 3). The portion of the cyst that extended under the pecotralis major muscle was accessed by placing additional 3 mm ports on the lateral border to retract the pectoralis minor muscle during the dissection. The lymphangioma was completely excised and the patient was discharged the same day with a drain in place. He was seen in follow-up and the drain was removed (Figure 2). He remains well without symptoms, complications, or recurrence at two months post-operative.
Preference Card
- 3mm and 5mm laparoscopic trocars
- 3mm and 5 mm laparoscopic instruments
- 5 mm 30 degree camera
- CO2 insufflation
- Bipolar sealing device
Tips and Pitfalls
- Because these cases are rare and individualized for each patient’s anatomy, preoperative planning plays an integral role in the success of the procedure. One must anticipate the orientation of the anatomy and the view prior to attempting such a procedure. For the planning of this case, the authors carefully reviewed minimally invasive literature within the area of interest, the cross-sectional imaging in multiple planes, as well as a digital anatomy program to anticipate the orientation of anatomic structures (3, 4, 5).
- One should have experience with camera-guided port insertion in order to develop expertise. This can be done on routine thoracoscopic or laparoscopic cases. One should also be comfortable with the appearance of a lymphangioma and how it differs from normal tissue prior to using this technique.
- The plane between the lymphangioma and normal tissue should be bluntly dissected prior to decompressing the cyst, as failure to do so will compromise further dissection as the CO2 insufflation will fill the cyst and make the dissection more challenging.
References
- Philippakis GE, Manoloudakis N, Marinakis A. A rare case of a giant cavernous lymphangioma of the chest wall and axilla in an adult patient. Int J Surg Case Rep 2013; 4(2): 164-166
- String A, Berber E, Foroutani A, Macho JR, Pearl JM, Siperstein AE. Use of the optical access trocar for safe and rapid entry in various laparoscopic procedures. Surg Endosc. 2001;15(6):570-3.
- Tolley N, Arora A, Palazzo F, et al. Robotic-assisted parathyroidectomy: A feasibility study. Otolaryngol Head Neck Surg 2011;144:859-866
- Tolley N, Garas G, Palazzo F, Prichard A, Chaidas K, Cox J, Darzi A, Arora A. A long-term prospective evaluation comparing robotic parathyroidectomy with minimally invasive openparathyroidectomy for primary hyperparathyroidism. Head Neck. 2014. Dec 24 [Epub ahead of print]
- Wu SD, Fan Y, Kong J, Yu H. Single incision for quadrantectomy and laparoscopic axillary lymph node dissection in the treatment of early breast cancer: initial experience of 5 cases. J Laparoscopic Adv Surg Tech A. 2014; 24(11):791-4







Comments