A minimally invasive approach to resection of early stage lung cancers has been described and many favorable results validated. There is increased interest in pursuing a robotic approach as early experience indicates some potential advantages, including less pain in the early postoperative period and possibly more complete hilar and mediastinal lymph node dissection.
Patient Selection
We find that peripheral tumors <5 cm are ideal to build a thoracic surgeon’s early experience and confidence on the robot. We typically have already performed a mediastinoscopy and have a tissue diagnosis before the OR precluding any need for a wedge intraoperatively. These two factors reduce the length of the procedure, facilitate the docking of the robot in an efficient manner, reduce concerns about local recurrence after wedge followed by completion lobectomy, and optimize appropriate staging before resection undertaken. In our early experience, we generally excluded central tumors, T3 or T4 tumors, N3 disease, patients needing sleeve resection, and those having undergone induction therapy. We do not exclude patients based on BMI, ASA status, or age. In fact larger BMI, older patients and higher ASA may benefit from a minimally invasive approach rather than a thoracotomy.
Operative Steps
Before patients are brought into the OR suite we turn the table around and use the foot as the head, with the addition of the head piece at this location. This allows the robot to dock unencumbered by the pedestal of the OR table.
 |
| Figure 1 |
 |
| Figure 2 |
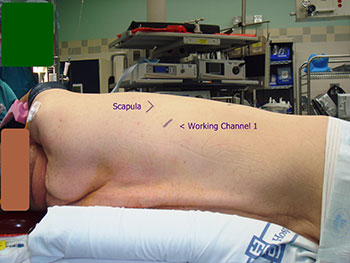
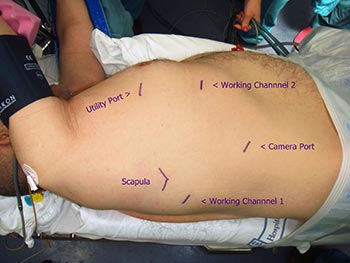
Patients are then brought into the OR and a double lumen is placed, followed by positioning in full lateral decubitus. The ipsilateral arm must be maintained horizontal to the shoulder or lower so as not to interfere with the docking or subsequent motion of the robot during the conduct of the operation, and should be positioned such that access to 4th interspace anteriorly is readily attainable. We also place extra long ventilation tubing to the anesthesia cart such that adequate space can be created between the anesthesiologist and the patient for docking of the robot. The drapes should be dropped and not hung to a pole. Figure 1 shows our preferred port placement. Figure 2 demonstrates that the hip of the patient is level with the thorax so as not to interfere with camera movement. We do not use epidurals for these cases, but liberally administer Toradol at conclusion of case.
We begin by dropping the ipsilateral lung and preemptively placing Marcaine in the interspace. All port sites receive Marcaine before port insertion. Access to the pleural space is first obtained using a hemostat to reduce nuisance bleeding. Our first site is low in the chest at the posterior axillary line, at a site determined by auscultation just above the diaphragm. We place a 12 mm trocar and use a 12 mm 0 degree scope initially. We insufflate CO2 for a few minutes only, at a preset pressure of 6 and with a flow of 6 L/min. Our next port is typically a posterior 8.5 mm port placed at least a hand breadth away from our camera site about 2 rib spaces inferior to the scapular tip, and 1 cm posteriorly. This incision must not be made too large as the robotic ports will then migrate in and out of the chest as tissue resistance is lost. However as the staplers are typically introduced into the chest via this posterior port, it is important that it is big enough so this maneuver happens without excessive trauma to the tissues. This must be balanced against the fact that posteriorly the rib space is tighter and therefore more painful postoperatively in our experience. The next port placed is the anterior 8.5 mm robotic port in the 7th intercostal space of the anterior axillary line. Lastly we place a 10 mm port in the 4th intercostal space of the anterior axillary line. This is used as a dynamic port for the assistant at the bedside. We generally introduce a suction catheter or ring forceps through this site, and the assistant controls all instruments and actions through this port.
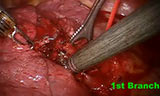
Next the robot is docked, with the boom coming in at an angle just behind the right ear, such that the camera angle is directed to the hilum and straight at truncus. At this point we switch to a 30 degree down 12 mm camera. We typically begin with a grasper in the right hand and a hook in the left hand. The inferior pulmonary ligament is initially taken down with exposure being aided via the bedside assistant through the most anterior/superior port. All nodes at station 9 are harvested. We then continue superiorly with the posterior hilar dissection, defining the crotch between the RUL bronchus and bronchus intermedius. All level 7 nodes are dissected and sent for analysis and the sump node is removed, which will facilitate later dissection of the PA as well as eventual stapling of the RUL bronchus. We continue our perihilar cut as far anterior as possible (Video Clip 1).
We then switch our exposure to the anterior hilum by flipping the lung posteriorly. We begin again with the grasper in the left arm port and the hook in the right to take down the pleura and expose the superior pulmonary vein and middle lobe branch. We continue the perihilar cut apically to join the posterior dissection already performed. Care is taken to avoid the phrenic nerve. We have found that using a grasper in the left hand and fenestrated bipolar in the right allows the safest backwall dissection and eventual stapler passage around vessels and bronchus. The superior pulmonary vein excluding the middle lobe vein is dissected and stapled with the stapler entering the chest from the posterior port after that arm is removed from the chest. There are lymph nodes around the truncus which lie just posterior and superiorly to the vein, and so it is most useful to remove those 10R nodes before stapling the vein as this allows safer passage of the stapler (Video Clip 2). The port is then reinserted into the chest. This is done most quickly and efficiently by simply placing the obturator into the port and with the port attached to the robot reinserting the whole unit back into the chest. Next the truncus is exposed and all level 10R nodes are removed before any passage of instruments is performed behind the PA. It is the dissection of the nodes around the PA that we believe is done more easily with the superior optics and instrumentation of the robot where some of the more important advantages of robotic surgery may be noted. Once all nodes are removed from the truncus, a stapler is then passed from usually the posterior port (sometimes the angle from the anterior port may be more favorable). We then reinsert whichever robotic port has been removed. It is not uncommon to take truncus trunks individually to ensure safer passage of the stapler.
Generally the last structure to be taken is the RUL bronchus. Much of the dissection has already been performed initially during the posterior exposure. In order to optimize passage of the stapler we may complete our fissures first and remove perihilar nodes. If the posterior recurrent branch is readily apparent it can be stapled before taking the bronchus. Partially or fully completing the fissure can help with passing of the stapler around the bronchus or artery (Video Clip 3). The lobe is left in the inferior hemithorax and additional mediastinal and hilar nodes are harvested, especially from levels 4R and 7. We then check for an air leak and ensure hemostasis.
We use an Endosac to remove the specimen. This is inserted from the anterior inferior port and the specimen is placed into the bag using instruments in the remaining two ports. After the Endosac is closed a long string is left on the bag and a hemostat is attached. The camera is then removed and the robot undocked. A grasper then is brought from the camera port anteriorly and the string is grasped and brought out the camera port. The camera incision is enlarged enough to remove the specimen. We then hold the camera by hand, ensure hemostasis, perform rib blocks, insert a chest tube apically, and reinflate the lung. If a Blake drain is placed it should be tunneled to avoid leakage of fluid or introduction of air upon its removal on the ward. All sites should be closed meticulously taking care to close the deeper and more superficial tissues as leakage of fluid can be a nuisance in the postoperative period and may require additional sutures on the ward or in the clinic after discharge. Toradol is administered if no contraindications exist and the patient is extubated in the operating room.
Preference Card
- Prograsp
- Maryland
- Monopolar shears
- Cautery hook
- Bipolar forceps
- Thoracic grasper
- Clip applier
- Endosac
Tips and Pitfalls
- It is important to include the OR staff and anesthesiology in initial operative phase planning for robotic lobectomy. Anesthesiology will most likely request face forward proximity to the patient airway for intraoperative dual lumen endotracheal tube management. The OR staff will need to pre-plan OR furniture arrangement with anesthesia requirements in mind.
- In addition to the anesthesia machine, relevant furniture includes the robot, OR bed, equipment boom, scope table, sterile back table, supply carts, monitors, scope warmer, and possibly bronchoscopy equipment.
- The OR bed itself should be turned 180°, headpiece to foot, with pedestal end towards the feet to accommodate unencumbered docking of the robot.
- Electrical cord management should ensure surgeons obstacle free movement from one side of the patient to the other and also back and forth to the robot console, in a darkened room, as needed.
- It is prudent to preplan for conversion to open lobectomy.
- Our OR robot team is a thoracic trained staff. Adjunct team members initially included an Intuitive trained staff member and vendor to ensure safe docking, routing and troubleshooting capabilities.It does take some time to get used to appropriate docking and undocking of the robot in timely fashion. In order to expedite the process it helps to have a nursing team that is consistent if possible. It may be necessary to turn the OR table around so the robot can gain enough access onto the patient and not be impeded by the pedestal of the table.
- The ventilator tubing should be long so the anesthesiologists have plenty of room, and the planed arm should afford access to the axilla as well as not be too high so it doesn’t impede docking of the robot.
- The ports should not be made too large or else they will slide excessively during the operation, and the posterior port should be made large enough to accommodate your stapler or else pain and/or bleeding can be troublesome.
- The subcutaneous access to the ribs should be straight and not tunneled for any ports. If tunneling has occurred this should be redone as it will limit the motion and freedom of that robotic arm.
- The angle of the robot for docking is crucial, and should be brought into place just behind the patient’s ear. The boom of the robot should be in direct line with the angle of the camera that you desire.
- We like to use an additional port anterior and superior for the bedside assistant to have access to the chest. We do not use any wound protectors or rib spreading at any time.
- We believe that lymph node dissection is superior with the robot and that this is the key to a safe operation. Once the nodes are dissected passing of the stapler is much safer.
- We try to have an experienced member of the team at the bedside to pass staplers around named structures of importance. We believe this to be just as important as a skilled surgeon on the console.
- We have not had to change the camera to any other port sites during the operation. It is possible for most lobes to do much of the dissection while avoiding fissure dissection, reducing air leaks and ultimately time in the hospital.
- Most structures of importance will be taken with the stapler entering the chest via the posterior site, which is also the narrowest rib space so this should be considered when choosing this site initially. When patients have needed narcotics for an extended period it has inevitably been due to pain in this posterior site.
- It is probably most safe to use the blunt instruments when dissecting the backside of structures, such as the Prograsp or fenestrated bipolar.
- We generally attempt not to grab the lobes that are remaining in the chest and retract by manipulating the lobe to be resected to reduce air leaks as well.
- We use the Endosac and bring the sac with the specimen out the camera port as we believe that patients have the least amount of pain with that incision. It is made just large enough to allow removal of the specimen.
- When closing the ports, it is crucial to close the subcutaneous tissues to reduce leakage of fluid since these ports were created to enter the chest in a direct fashion and have not been tunneled. If this is not done the patient will return with fluid leak and may require additional sutures on the ward or in the clinic.
- Rib blocks should be performed before leaving the OR.
Results
In the last year we have been able to successfully complete all robotic anatomic lung resections that were not after induction therapy or needed a chest wall resection. Operative time, blood loss, and length of stay were similar to VATS, but increased nodes were harvested with the robotic approach and less narcotics were needed by patients, with many off all narcotics by the first clinic visit. In general we performed robotic surgery on older, more frail patients with higher ASA and have had no operative mortality.
References
- Veronesi G, Galetta D, Maisonneuve P, Melfi F, Schmid RA, Borri A, Vannucci F, Spaggiari L. Four-arm robotic lobectomy for the treatment of early-stage lung cancer. J Thorac Cardiovasc Surg. 2010 Jul;140(1):19-25. Epub 2009 Dec 28.
- Gharagozloo F, Margolis M, Tempesta B, Strother E, Najam F. Robot-assisted lobectomy for early-stage lung cancer: report of 100 consecutive cases. Ann Thorac Surg. 2009 Aug;88(2):380-4.
- Gharagozloo F, Margolis M, Tempesta B. Robot-assisted thoracoscopic lobectomy for early-stage lung cancer. Ann Thorac Surg. 2008 Jun;85(6):1880-5; discussion 1885-6.
- Park BJ, Flores RM, Rusch VW. Robotic assistance for video-assisted thoracic surgical lobectomy: technique and initial results. J Thorac Cardiovasc Surg. 2006 Jan;131(1):54-9.