Traumatic rupture of the thoracic aorta is a common cause of death after motor vehicle accidents. The gold standard for treatment of traumatic rupture of the descending thoracic aorta is a left thoraoctomy with replacement of the affected aorta. The endovascular treatment of aortic transection is an emerging alternative in multi-traumatized patients. This case presents the utility of intravascular ultrasound (IVUS) in endovascular treatment of a contained rupture of descending thoracic aorta.
Patient Presentation
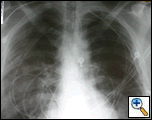
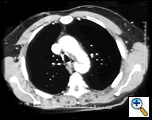
A 44 year old white male with 40 pack-year history of smoking was driving his motor cycle when a car struck him from the left side at approximately 40 mph. The patient lost consciousness at the scene, but upon arrival to trauma bay, he was alert and oriented. He complained of shortness of breath and pain in the left chest and left lower and upper extremity. The trauma team at the referring institution initiated advanced trauma life support pathway. He was found to have left femur, humerus, tibial-fibular and a complex pelvis fractures. The initial chest x-ray revealed a widened mediastinum (Figure 1), and a high-resolution contrast-enhanced computed tomography demonstrated proximal descending aortic rupture with hemomediastinum (Figure 2), left pulmonary contusion and a small left hemopneumothorax.
The patient remained hemodynamically stable at the outside hospital, and was evaluated by the local cardiothoracic surgeon for open repair. Secondary to his significant injuries, he was felt not to be a candidate for cardiopulmonary bypass and hypothermic circulatory arrest. After discussing the risks and benefits of an endovascular repair, the patient agreed to proceed with the endovascular option and was transferred to Harbor-UCLA Medical Center 15 hours after the accident..
Informed consent was obtained and the patient was offered endovascular repair of the contained aortic rupture using the Medtronic Talent endoprosthesis through a single institution Investigator Investigational Device Exemption (IDE) approved by the FDA. In the endovascular suite, the patient was placed in supine position and underwent local anesthesia with sedation. The left common femoral artery was exposed and then accessed using an 18-gauge needle and a 0.035 inch flexible-tipped Bentson Starter guidewire was passed in a retrograde fashion into the descending aorta under fluoroscopic guidance. 3000U of Heparin was administered and a 14-Fr introducer sheath was placed in the left common femoral artery. The IVUS (Volcano Visions, PV 8.2 F) catheter was used to guide and advance the Starter guidewire safely across the contained rupture area into ascending aorta. Upon advancement of the IVUS probe into the thoracic aorta, the area of transection was encountered. There was a dissection flap with a dilated and disrupted area in the middle of aorta
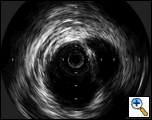 |
| Figure 3: IVUS showing the site of aortic rupture. |
(Figure 3). The proximal aspect of the transection was 1.5 cm below the origin of the left subclavian artery. Origins of the arch vessels were marked on the masked picture on the fluoroscopy monitor. Using the IVUS, the aortic diameter was measured 28 mm just distal to the left subclavian artery and 29 mm at origin of the left common carotid artery. It was decided to cover the left subclavian artery to get an adequate landing zone for the endoprosthesis. Therefore, the IVUS was placed just distal to the left carotid artery and its position marked on the fluoroscopic screen. The catheter was then withdrawn until above, and then below the transected area. These measured 36 mm and 76 mm in length, respectively. With these measurements in mind, a 30 mm diameter by 130 mm in length Medtronic Talent device with 15 mm proximal wire configuration was selected to exclude the transected portion of the aorta.
Subsequently, the right radial arterial line was exchanged to a 4F Pinnacle introducer. A 180 cm angled tip Glidewire was inserted into the introducer, and it was advanced over the innominate artery into the aortic arch. A 4F Pigtail catheter was inserted over the guidewire into the ascending aorta. The left femoral Bentson Starter guidewire was exchanged for a 0.025in x 260cm Platinum Plus ST guidewire. The introducer sheath was removed from the groin and the Medtronic Talent endoprosthesis was inserted over the stiff guidewire. The external and common iliac arteries were found earlier to be suitable for this device on the IVUS examination (7 and 8 mm, respectively). The device was maneuvered into previously marked position and an aortogram was performed to confirm correct graft placement. Adenosine-induced cardiac arrest was not utilized in this patient due to
 |
| Figure 4: “Completion”-IVUS examination showing exclusion if the proximal descending aorta. |
concerns about myocardial contusion. After lowering the systolic blood pressure to 100 mmHg, the stent graft was deployed precisely distal to the left common carotid artery. Small boluses of contrast were administered during deployment to confirm device position relative to the left carotid artery. A completion aortogram confirmed adequate exclusion of the transected area and no evidence of an endoleak was identified. A completion IVUS exam was performed, showing complete apposition of the device to the aortic wall circumference (Figure 4). Subsequently, the catheters and the wire were removed from the left groin, and the left common femoral artery is repaired under direct vision. The patient was found to have bounding distal pulses and was neurologically intact after the procedure. The pulses of the left arm were detectable with Doppler. Although the systolic blood pressure in the left arm decreased by 30 mm Hg, the patient did not demonstrate any ischemia or claudication symptoms.
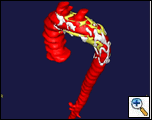 |
| Figure 5: Post-deployment three dimensional reconstruction of contrast-enhanced computer tomography study. The left subclavian artery is filling in a retrograde fashion. |
The patient was transferred to Intensive Care Unit, and underwent repair of his orthopedic fractures the next few days. On post-operative day two, a contrast-enhanced high-resolution computer tomography scan was obtained. Figure 5 is the three-dimension reconstruction of this study using the software of Medical Metrx Solutions, Inc. (West Lebanon, NH). It demonstrates an excluded transected aorta without evidence of endoleak. Retrograde blood flow to the left subclavian is present on this study. Following several orthopedic procedures, patient was discharged to home two weeks later.
Comments
Improvement and sophistication of operative techniques has led to better outcome in patients undergoing aortic surgery. However, poly-traumatized patients presenting with a contained aortic rupture are often not surgical candidates for anticoagulation and cardiopulmonary bypass that would be necessary for open surgical repair of this aortic pathology. Furthermore, many older patients may not have significant cardiopulmonary reserve necessary for traditional operation, thus making them high surgical risk patients. Thoracic endovascular aortic repair (EVAR) of contained aortic rupture is a promising alternative to traditional open surgical repair. Given the high morbidity and mortality in the patients that are treated with open repair, it is likely that EVAR will become the standard treatment for such pathology in near future. Data from prospective multi-center trial using the GORE TAG endoprosthesis suggests that endoluminal grafting for descending aortic aneurysms is associated with decreased length of stay, less blood transfusion and earlier return to a baseline activity lifestyle compared with open repair [1]. Although endoluminal treatment of descending thoracic aorta aneurysm is the only FDA-approved indication for the TAG endoprosthesis, EVAR of acute and chronic type B dissection, penetrating aortic ulcers, aortobronchial fistulas as well as contained aortic ruptures are treated using thoracic endoluminal devices in few medical centers [2-5]. Most of these centers have a Single-Center Investigator IDE to use the thoracic endoprosthesis for non-FDA approved indication. The Medtronic Talent Device, that was used in this case, has not yet received FDA approval. The results of multi-center Valor Trial has completed enrollment, and efficacy and safety of device for exclusion of descending thoracic aorta aneurysms is pending.
EVAR in this patient was performed using IVUS. Ultrasound is the most accurate modality for measuring any vessel’s diameter and facilitates the correct sizing of landing zone and proper selection of the device. Furthermore, it helps identifying ideal location for fixation points, by showing plaque morphology, calcium & thrombus at the proximal and distal landing zones. IVUS significantly reduces the need and the amount of contrast media. In fact, this case could have been safely performed without any contrast material. This is crucial in patients with multiple co-morbidities such as acute or chronic renal insufficiency. Furthermore, IVUS significantly reduces total fluoroscopy exposure to the patient (less than six minutes in this patient). This is of importance when considering the accumulative dose of radiation that these patients will be exposed in the long-term follow-up.
IVUS will change the paradigm of management of patients with contained aortic rupture. Since contrast-enhanced computer tomography is not always diagnostic in trauma patients, the gold standard has been to perform a time-consuming aortogram. Under the proposed paradigm, the patient suspected to have aortic rupture, may be taken immediately after the computer tomography to the operating room with endovascular capabilities. An IVUS examination will make the definitive diagnosis, and the patient may receive immediate treatment for his/her aortic injury. This is of utmost importance in unstable patients. If the mechanism of injury, the patient’s presentation to trauma bay, or the first chest x-ray are suggestive of an aortic disruption, and there is no other source for patient’s hypotension, this patient may be taken immediately to the operating room with endovascular capabilities for diagnosis and endoluminal repair. Furthermore, in patients with unexplained hypotension and widened mediastinum on chest X-ray, patient may undergo laparatomy in the operating room while having an IVUS examination of descending aorta through the femoral vessels. IVUS will penetrate the field of endovascular, general and cardiovascular surgery- as it has done the field of interventional cardiology. For the aforementioned reasons, it is crucial for future cardiovascular surgeons to embrace this technology.
References
- Makaroun,MS, Dillavou,ED, Kee,ST, et al. Endovascular treatment of thoracic aortic aneurysms: results of the phase II multicenter trial of the GORE TAG thoracic endoprosthesis. J Vasc Surg 2005;41:1-9.
- White RA, Donayre CE. Walot I., et al. Endovascular exclusion of descending thoracic aortic aneurysms and chronic dissections: Initial clinical results with the AneuRx device. J Vasc Surg 2001;33:927-34.
- Criado FJ. Abul-Khoudoud, OR, Domer GS, et al. Endovascular repair of the thoracic aorta: lessons learned. Ann Thorac Surg 2005;80:857-63.
- Greenberg RK, O'Neill S, Walker E. et al. Endovascular repair of thoracic aortic lesions with the Zenith TX1 and TX2 thoracic grafts: intermediate-term results. J Vasc Surg 2005;41:589-96.
- Donayre CE. Khoynezhad A. Bui H., et al. Spinal cord ischemia following endovascular repair of descending thoracic aneurysms and dissections. J Vasc Surg (in press).