Patient Selection
The intercostal muscle (ICM) flap has proven to be a reliable buttress for the bronchial stump following pneumonectomy and lobectomy. (1-3) It has also been used to reinforce suture lines after bronchoplastic resections, esophageal repair and esophagogastric anastomoses. (4, 5) Recent evidence supports the use of an ICM flap after lung resection in high risk patients. These include patients with poorly controlled diabetes, pulmonary infections, immunocompromised patients, those receiving neoadjuvant therapy or adjuvant radiation (N2 disease found at the time of surgery) or chemotherapy. (1-3) In addition, creating an ICM has been associated with less pain and quicker return to function following lung resection with thoracotomy. (6-8) There has been only one report of video assisted thoracoscopic (VATS) mobilization of ICM flap in the literature.(9) The technique described below allows harvest of an intercostal flap in patients undergoing thoracoscopic lobectomy.
No additional preoperative workup is needed for the procedure other than that required for the underlying condition. One of the main advantages of using an ICM flap is that it is a well vascularized structure. However, if the vascular pedicle has been injured and its viability is of concern, then another flap should be considered for buttressing. Previous thoracotomy in the same intercostal space of the working incision would make that ICM difficult to harvest for use as a buttress. Standard contraindications to VATS would prevent the use of this technique in harvesting the intercostal muscle.
Operative Steps
The patient is placed in the standard lateral decubitus position for a posterior thoracotomy. The arm is positioned on an airplane to ensure that it will not interfere with the use of the instruments. The surgeon stands anteriorly while the assistant can stand on either side of the patient.
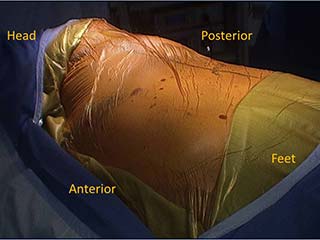 |
| Figure 1. Camera port and working incision. |
The camera port is placed slightly posterior to the anterior superior iliac spine line in the 7th or 8th intercostal space. The working incision is placed in line with the major fissure usually in the 5th intercostal space in the mid axillary line (Figure 1). The muscles of the chest wall are divided carefully with cautery until the intercostal space is reached. Retraction using army-navy retractors in the superior and inferior aspect of the wound help with exposure. The intercostal space is identified when the rib cage is reached and using a finger, blunt dissection is performed posteriorly and anteriorly to separate the other muscles of the chest wall from the intercostal space. Low cautery settings are used to decrease the risk of thermal injury to the neurovascular pedicle. An incision is made in the intercostal muscle where it attaches to the lower rib for the length of the working incision. The intercostal muscle is then detached from the superior aspect of the lower rib after entering the pleural cavity using a combination of cautery and blunt dissection. This can be done gently with a periosteal elevator or a peanut dissector. An army-navy or Richardson retractor can be used to retract the anterior aspect of the incision to allow for mobilization greater than the length of the working incision. This is also done posteriorly. The superior aspect of the flap is left attached to the under surface of the upper rib in the intercostal space at this time to avoid potential damage to the vascular pedicle of the flap from passage of instruments through the working incision during the operation. However, when the intercostal space is narrow, mobilization of both superior and inferior aspects of the flap can be performed initially to allow more room for passage of instruments through the space and less trauma to the flap. Dissection of the superior aspect of the flap is done by incising the intercostal muscle where it attaches to the inferior edge of the upper rib in the intercostal space. The muscle is detached from the under surface of the rib using a combination of blunt and cautery dissection. The blade of the cautery is used for blunt dissection while inferior traction is placed on the muscle using a Debakey forceps or peanut dissector. The pleural cavity is entered early as this enables traction on the flap using a finger and allowing for the parietal pleura of the flap to be divided first allowing an easier and safer dissection of the vascular pedicle. Complete mobilization of the ICM is not performed initially because it can obstruct the view and hinder dissection during the pulmonary resection. There is no retraction of the intercostal space that would injure the flap during VATS lobectomy, therefore, there is little benefit in harvesting the entire flap prior to the lung resection.
The planned resection is then performed and the bronchial stump checked for an air leak.
A tonsil sponge in a thoracoscopic ring clamp is then used to bluntly dissect the ICM from the serratus anterior, the latissimus muscle and other soft tissues of the chest wall posteriorly. This is facilitated by elevating the cut edge of the chest wall musculature with an army navy retractor from the ICM in the working incision and passing the tonsil sponge stick in this plane posteriorly. This is an important step of the operation. Without this maneuver, the intercostal flap will remain attached to the chest wall even after dissection from its costal attachments and detaching it from the chest becomes difficult. This posterior blunt dissection is carried back to the spine.
The superior aspect of the ICM is dissected from the inferior aspect of the superior rib in the intercostal space as described above (Video 1a below). This is done under direct vision in the space of the working incision. The soft tissues can be retracted with army-navy retractors or a Weitlaner self retaining retractor. The ICM is then dissected anteriorly under either direct vision or with the thoracoscope detaching both sides of the muscle from the ribs until the intercostal space approaches the mediastinum, i.e. the pericardium (Video 1b below).
The posterior dissection of the ICM is then performed, starting under direct vision with retraction of the muscles of the chest wall with a Richardson or army navy retractor. Either the superior or inferior aspect of the muscle can be dissected first. Once dissection under direct vision is not possible then the thoracoscope is used to provide visualization through the working incision. At this stage the retraction of the muscles of the chest still facilitates the dissection of the ICM and should be continued. The cautery is changed to a “long” blade and the ICM is retracted with a peanut dissector as the dissection is continued posteriorly in the intercostal through the working incision (Video 2 below). The suction can also be used to retract the ICM and helps provide a clearer operative field. This part of the mobilization is the most challenging as the camera and instruments are placed through the same incision and patience is required until the assistant and operator gain experience. Once the ICM has been dissected back to the scapula the camera can be placed in the camera port for further visualization of the dissection. Retraction of the chest wall is also no longer needed. The operator continues to use the peanut dissector for retraction of the ICM and a combination of cautery and blunt dissection (Video 3 below). The superior and inferior parietal pleura of the intercostal space are divided posteriorly to the level of the neck of the rib. The superior aspect of the intercostal space is then dissected approaching from the pleural side of the chest (Video 4 below). Visualization with the scope is important to ensure the pedicle of the flap is preserved. Keeping the plane of dissection as close to the rib as possible also prevents injury to the pedicle. Next, the inferior aspect of the ICM is detached from the rib. This allows the ICM flap to hang down into the chest if the blunt dissection of the chest wall muscles from the ICM has been completed. If not, further blunt or cautery dissection must be performed to free the ICM from the chest wall.
Once dissection of the ICM has been completed posteriorly to the neck of the rib, there should be sufficient length to the flap to permit it to reach the bronchial stump easily and without tension (Video 5 below). The muscle can either be divided sharply anteriorly or left in situ and positioned over the bronchial stump making sure the muscle is not twisted (Video 6 below). If the muscle is divided, the anterior end of the ICM stump is ligated with 2-0 silk. 4-0 Vicryl sutures in an interrupted fashion are then used to secure the ICM to the bronchial stump while avoiding injury to the neurovascular bundle (Video 7 below). This can be done using a thoracoscopic or long needle driver and a pick up through the working incision. Knots can be tied intra- or extracorporally (utilizing a knot pusher or right angle clamp) as per the skill of the operator. It is possible to tie the knot in an open fashion with the operator’s finger being able to reach the bronchial stump through the working incision (Videos 8a, 8b below). The sutures can be placed through the pleura of the ICM and then the bronchus just adjacent to the staple line on the superior and inferior aspect of both structures draping the ICM over bronchial staple line. Alternatively, the ICM can be sutured directly to the bronchial staple line. The end result should be a completely covered bronchial stump with a viable portion of the ICM (Video 9 below). If the ICM has been divided anteriorly, then securing the tip of the muscle to the stump should be avoided as it is the most likely area to be ischemic. An area 4-5 cm distal to the tip of the flap should be used. Two additional sutures are placed posterior to the stump and two anterior to secure the muscle to the mediastinal pleura ensuring complete coverage of the bronchial stump and decreasing the risk of avulsion of the ICM from the bronchus.
Once this is completed the bronchial stump is checked again for an air leak and the chest cavity is inspected for hemostasis. The ICM is carefully observed for any bleeding points. Gentle pressure, 5mm clips or cautery on a low setting can be used to control the bleeding.
The chest tube is placed posterior to the ICM flap to decrease the chance of compression of the neurovascular bundle when the lung is ventilated (Video 10 below). The flap is observed when the lung is re-inflated to ensure it is not dislodged or is placed under tension by the expanded lung. The working incision can be closed by approximating the muscles of the chest wall. Additionally, two posteriorly placed pericostal or intracostal (holes drilled in the lower rib with sutures placed through these holes and around the upper rib) sutures of number-2 vicryl can be used to approximate the intercostal space to prevent potential lung herniation.
Preference Card
- Thirty degree scope
- Insulated cautery tip with extender
- Thoracoscopic tonsil sponge stick
- Army-Navy retractor
- Peanut dissector
- Thoracoscopic sucker
- Thoracoscopic or standard needle holder
- Right angle clamp or knot pusher
Tips and Pitfalls
- A tonsil sponge stick is used to separate the serratus anterior and latissimus muscles from the chest wall and ICM prior to mobilization of the ICM
- Cautery setting is kept low (10-20) when harvesting the muscle flap to limit thermal injury to the neurovascular bundle
- Initial mobilization of the ICM can be done through the working port
- The anterior aspect of the ICM can be divided or left in situ
- Mobilize the ICM posteriorly to the neck of the rib and anteriorly to the mediastinum.
- Do not use the tip of the ICM to buttress the suture or staple line
- Secure the ICM to the bronchus and the mediastinal pleura
- Check the ICM for areas of bleeding
Results
Developing a bronchopulmonary fistula (BPF) following pulmonary resection can be a disastrous complication. The ICM has been shown to be a reliable buttress following pulmonary resection in high risk patients. (1-3) Cerfolio et al reported no BPFs with ICM buttressing after 152 lobectomies in patients who received preoperative chemoradiation therapy.(3) In 68 diabetic patients who underwent a pneumonectomy, 35 had a ICM buttress of the bronchial stump while 33 patients did not. No BPFs or postpneumonectomy empyemas were noted in the group that received ICM buttressing compared to 6 (8.8%) and 5 (7.4%), respectively, in those patients who did not have an ICM buttress. (2) The incidence of BPF following VATS lobectomy has yet to be determined. After 1,100 VATS lobectomies, McKenna et al reported only 3 BPFs. (10)
We started performing this procedure after 2 BPFs occurred following VATS lobectomy in patients with poorly controlled diabetes who received adjuvant chemotherapy. We have successfully performed this procedure in 12 patients, all of whom had diabetes mellitus, and have had no BPFs. Initially we divided the ICM anteriorly, but the last 6 we have not divided the ICM. This allows the muscle to maintain its natural orientation and is easier to handle the muscle within the chest and position it over the bronchus. As the anterior blood supply of the ICM is preserved, this may result in a better vascularized flap. There is evidence that pain is decreased in patients in whom an ICM is mobilized when a thoracotomy is performed (7) and therefore this is unlikely to increase the pain of the VATS incision. One patient had post operative bleed related to the ICM which required re-operation and clipping of a branch of the intercostal artery. Whether harvesting an ICM will increase the incidence of intercostal hernia is unknown at this time.
Traditionally, harvesting of the ICM flap has been done following thoracotomy and has only been described once in the literature using VATS. (9) Our article describes in detail the technique, with associated images and video, to harvesting the ICM flap using VATS with minimal added risk to the patient and should be performed for the same indications following pulmonary resection with thoracotomy.
Video 1a. Mobilizing the superior aspect of the ICM thorough the working incision.
Video 1b. Mobilizing the ICM anteriorly using the thoracoscope.
Video 2. Mobilizing ICM through the working incision.
Video 3. Mobilization of the inferior aspect of the ICM.
Video 4. Mobilization of the superior aspect of the ICM.
Video 5. Full length of ICM flap.
Video 6. Anterior division of the muscle flap.
Video 7.Suturing the muscle flap to the bronchial stump.
Video 8a. Knot tying.
Video 8b. Knot tying using a right angle clamp.
Video 9. Completely covered bronchial stump.
Video 10. Positioning of the chest tube.
References
- Maniwa T, Saito Y, Kaneda H, Imamura H. Bronchial stump reinforcement with the intercostal muscle flap without adverse effects. Eur J Cardiothorac Surg 2006;30(4):652-6
- Sfyridis PG, Kapetanakis EI, Baltayiannis NE, et al. Bronchial stump buttressing with an intercostal muscle flap in diabetic patients. Ann Thorac Surg 2007;84(3):967-71.
- Cerfolio RJ, Bryant AS, Jones VL, Cerfolio RM. Pulmonary resection after concurrent chemotherapy and high dose (60Gy) radiation for non-small cell lung cancer is safe and may provide increased survival. Eur J Cardiothorac Surg 2009;35(4):718-23.
- Rendina EA, Venuta F, Ricci P, et al. Protection and revascularization of bronchial anastomoses by the intercostal pedicle flap. J Thorac Cardiovasc Surg 1994;107(5):1251-4.
- Cerfolio RJ, Bryant AS, Yamamuro M. Intercostal muscle flap to buttress the bronchus at risk and the thoracic esophageal-gastric anastomosis. Ann Thorac Surg 2005 Sep;80(3):1017-20.
- Cerfolio RJ, Bryant AS, Patel B, Bartolucci AA. Intercostal muscle flap reduces the pain of thoracotomy: a prospective randomized trial. J Thorac Cardiovasc Surg 2005;130(4):987-93.
- Cerfolio RJ, Bryant AS, Maniscalco LM. A nondivided intercostal muscle flap further reduces pain of thoracotomy: a prospective randomized trial. Ann Thorac Surg 2008;85(6):1901-7.
- Allama AM. Intercostal muscle flap for decreasing pain after thoracotomy: a prospective randomized trial. Ann Thorac Surg 2010;89(1):195-9.
- Sagawa M, Sugita M, Takeda Y, Toga H, Sakuma T. Video-assisted bronchial stump reinforcement with an intercostal muscle flap. Ann Thorac Surg 2004;78(6):2165-6.
- McKenna RJ Jr, Houck W, Fuller CB. Video-assisted thoracic surgery lobectomy: experience with 1,100 cases. Ann Thorac Surg 2006;81(2):421-6.