ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Bronchial Thermoplasty for Severe Asthma
Bronchial thermoplasty (BT) is a novel treatment option for uncontrolled severe asthma that targets the hypertrophied smooth muscle in the airways, which is considered to be part of the pathophysiology of and contributor to asthma symptoms. In May 2010, the FDA approved BT in patients 18 years and older, whose asthma is not well controlled with inhaled corticosteroids and long-acting beta agonists.
Indications
In general, patients with airway remodeling and those with uncontrolled asthma, in spite of being treated intensely at step 5 or 6 by NAEPP guidelines, are most appropriate for this treatment option (1). Patient selection must exclude asthma mimetics such as ABPA, COPD, vocal cord dysfunction, Churg-Strauss syndrome, drug reactions, congestive heart failure, bronchiolitis obliterans, chronic sinus problems, severe GERD, excessive dynamic airway collapse, and tracheobronchomalacia (Table 1).
- Documented adult asthma (with reversible decrease in FEV1 or positive methacholine challenge test)
- Uncontrolled asthma with medications (inhaled steroid > 500 mcg per day and/or LABA > 100 mcg per day)
- Pre-bronchodilator FEV1 equal to or greater than 60% predicted
- Uncontrolled but stable asthma without current URI, recent (in last 2 weeks) asthma exacerbation, and FEV1 is within 10% of the baseline value
- Non-smoker for 1 year or more (if former smoker, < 10 packs years)
- Those without active comorbidities that may aggravate asthma symptoms (see text)
Table 1. Indications for bronchothermoplasty
Contraindications and Complications
Table 2 shows typical contraindications. The main complications of bronchial thermoplasty are asthma exacerbation, atelectasis, and hemoptysis.
- General contraindications for flexible bronchoscopy (22)
- Presence of pacemaker, defibrillator, or other implantable devices
- Prior treatment with the Alair System
- Active respiratory tract infection
- Asthma exacerbation in the past 6 weeks
- Change in dose of systemic steroids (up or down) in last 2 weeks
- Known coagulopathy
- Those with comorbidities untreated (see text)
Table 2. Contraindications to bronchothermoplasty
Mechanism of Action
Delivering controlled heating to the airway wall causes a decrease in the amount of airway smooth muscle. Bronchial thermoplasty also reduces the ability of the airway to bronchoconstrict in response to noxious stimuli, such as methacholine. In most cases, there will be no improvement in FEV1, but bronchial thermoplasty will possibly cause less bronchospasm due to decreased muscle bulk in the airways (2, 3).
Technology and Equipment
The Alair Bronchial Thermoplasty System (Asthmatx, Inc., Sunnyvale, CA) delivers thermal energy in a controlled manner to the airway wall, aiming to reduce the airway smooth muscle mass. The system is comprised of two primary components. The first component is the bronchial thermoplasty single-use catheter, which can be used with a standard bronchoscope with a 2mm working channel. The catheter shaft is 1.4 mm and the expandable electrode arrays with a handle (4 arrays and 5 mm long) can be expanded to 10-13 mm in diameter, which is the maximum diameter of the airways that can be treated (Figure 1). The second component is the bronchial thermoplasty radiofrequency controller, which is designed with set control parameters and algorithms to deliver the correct intensity and duration of thermal energy each time (Figure 2). The controller has a foot switch pedal designed to deliver heat energy at 65°C on each application. In order to deliver the preset time and energy, all 4 arrays of the catheter should be gently touching the airway mucosa. The controller is also attached to the patient with a patient return electrode to complete the return path for the electrical current.

Figure 1. Alair Bronchial thermoplasty catheter
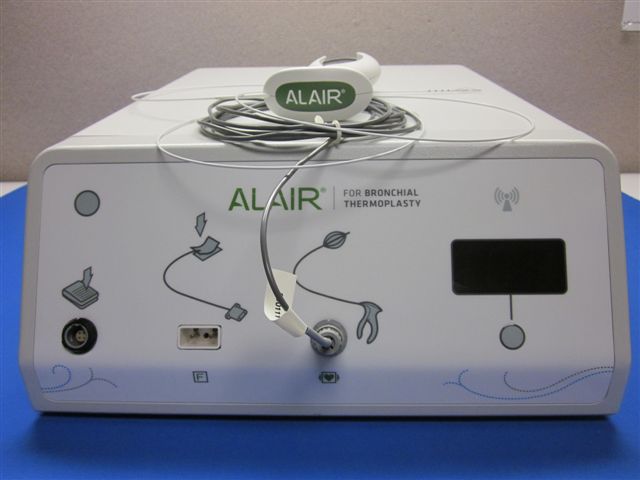
Figure 2. Bronchial thermoplasty radiofrequency controller
Procedure, Patient Preparation, and Post-Procedure Care
The patient should be stable enough for conscious sedation and bronchoscopy. Bronchial thermoplasty should be postponed if the patient was not premeditated with prednisone, the pulse oximeter is less than 90% on room air, the patient has increased asthma symptoms within the last 2 days, the patient has experienced a recent asthma exacerbation or a change in the dose of systemic corticosteroids in the past 14 days, the patient has an active respiratory tract infection, active allergies or sinusitis, or if there is other clinical instability. The procedure is performed in three separate sessions of bronchoscopy, separated at least three weeks apart.
In each session, one particular lung area is treated. Usually, the right lower lobe in the first session, the left lower lobe in the second session, and upper lobes in the last session. The right middle lobe is not treated because of the narrow opening of the lobar bronchus and acute take off angle where treatment related inflammation may lead to lobe collapse. After the patient is sedated, or is under general anesthesia, and the return electrode is placed, the bronchoscope is advanced to the target area and the treatment of each segment and subsegment is carried out in a systematic way. Because there may not be any visible effect on the mucosa, the treatment should be performed in a systematic manner distal to the proximal airways, without overlapping the treatment areas. The catheter is advanced distally to the level that the bronchoscopist is able to see the electrode arrays, which usually corresponds to 3 mm airways. After the smallest/distal area is treated, the catheter is pulled back 5 mm each time to treat the entire airway. This is done as long as all four electrodes in the array touch the airway wall mucosa, which is usually the airways measuring 13 mm in diameter. Each treatment application, which is achieved by stepping on the footswitch once, lasts 10 seconds. When the catheter handle is used to expand the electrode arrays, the arrays should only be touching the mucosa gently and should be released/relaxed between treatments.
Once the treatment session is completed, the patient is usually monitored 2-4 hours in the recovery area. Spirometry should be performed prior to discharge and the FEV1 should be within the 80% of the pre-procedure value. If not, overnight admission for observation is suggested.
References
- Expert panel report 3: guidelines for the diagnosis and management of asthma. Bethesda, MD: National Heart, Lung, and Blood Institute, August 2007. (Accessed on June 10, 2012).
- Cox PG , Miller J , Mitzner W , Leff AR . Radiofrequency ablation of airway smooth muscle for sustained treatment of asthma: preliminary investigations. Eur Respir J . 2004 ; 24 ( 4 ):659 - 663.
- Miller JD , Cox G , Vincic L , Lombard CM , Loomas BE , Danek CJ . A prospective feasibility study of bronchial thermoplasty in the human airway. Chest . 2005 ; 127 ( 6 ):1999.




