ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Direct Anatomic Reimplantation of Malignant Right Coronary Artery Without Aortotomy: A Two Patient Case Report
O’Connell D, Tendulkar A. Direct Anatomic Reimplantation of Malignant Right Coronary Artery Without Aortotomy: A Two Patient Case Report. October 2021. doi:10.25373/ctsnet.16887922
Background
Historically, direct reimplantation is a technique that requires a partial aortotomy and xeno-pericardial patch (1). A recent report outlined a simplified approach for the reimplantation of a right anomalous aortic origin of a coronary artery (AAOCA) that outlined a skeletonized technique that employs the principles of a standard aortic anastomosis in coronary artery bypass grafting (2). This technique mobilizes the coronary artery's proximal end by its removal from the aortic sinus. The excised hole can be repaired by a simple suture closure. Once the proximal vessel is mobilized, it can be reimplanted into the usual ostial sinus after punch biopsy creates a new ostium. This approach has been successful in our hands and others with modifications (3). Here, we present two cases using this approach for malignant right coronary arteries that arise from the left (anterior) aortic sinus with partial intramural segments and have a malignant course between the pulmonary trunk and aorta.
Cases
Patient 1: a 63-year-old obese Hispanic male with anginal symptoms and a history of PAD, cardiomegaly, GERD, HTN, and dyslipidemia.
Patient 2: a 61-year-old obese Hispanic male presented with anginal symptoms with a history of coronary artery disease and diabetes.
The workup for each patient began with a Lexiscan SPECT MPI stress test that showed mild inferior ischemia. Subsequent coronary artery angiograms depicted an anomalous right coronary artery originating from the left cusp superior to the left main artery take off. The malignant courses were confirmed with a coronary artery CT scan with contrast and 3D construction. A review of the coronary artery CT scan showed a malignant course of the RCA between the aortic sinus and the main pulmonary trunk, with narrowing and compression noted proximally (patient 1 imaging Figure 1a, patient 2 imaging Figure 1c). Varying partial intramural segments were also noted on CT for both patients. With the increased risk of further ischemia and shared decision-making, the patients opted to undergo open-heart surgery for reimplantation of the RCA to a trouble-free origin at the right (posterior) aortic sinus.
Operation and Recovery
The two patients underwent similar reimplantation operations, both with uneventful intraoperative complications. An example procedure is outlined below, along with postoperative considerations. The surgical procedure was performed with a routine median sternotomy and pericardiotomy. Next, identification of the anomalous right coronary origin was performed with low-energy cautery and sharp dissection. Once the field was adequately exposed, heparin was given. After confirmation of activated coagulation time (ACT; 480 seconds), routine cannulation was performed, and cardiopulmonary bypass initiated. While keeping the patient warm and the heart beating, the origin of the RCA was circumferentially exposed along the right ventricle towards the right atrioventricular junction. Once adequate length was mobilized under mild hypothermia of 34 degrees C, an aortic cross-clamp was placed and cardioplegic arrest achieved using antegrade and retrograde cannulae. At this point, the RCA's most proximal end was sharply dissected as close to the aortic sinus as possible. The aortic ostial end was then closed with 6-0 Prolene in running fashion. The root was briefly filled with cardioplegia and heart filled to mark the right aortic sinus for the appropriate tension-free reimplantation location. A 5-mm diameter punch aortotomy was performed at the marked site. 6-0 Prolene suture was used to carry out a direct anastomosis of the proximal free end right coronary artery to the aorta. The patient underwent routine weaning from cardiopulmonary bypass with reversal of heparin with protamine. Perfusion of the RCA was confirmed using flow probe, which exhibited excellent flow with minimal pulsatile index (patient 1 mean coronary flow measured 53ml/min while partially on bypass with a pulsatility index of 0.8, patient 2 mean coronary flow measured 100ml/min with a pulsatility index of less than 2.5). Routine sternotomy closure was performed. In the immediate postoperative course, the patients recovered well with no complications. Both patients were extubated 2-3.5 hours after arriving in the ICU. Patient 2 exhibited ST segment elevation on telemetry in the ICU that was not validated by multiple serial 12-lead EKGs. On postoperative day 2, both patients were transferred to the step-down unit and were discharged home on postoperative day 4. The alleviation of the malignant RCA course is shown with CT angiography at three months follow up (patient 1: Figure 1b, patient 2: Figure 1d). Since the procedure, respectively 9 and 14 months ago, neither patient has reported anginal symptoms and are readmission free.
Discussion
As Izumi et al. previously described, this technique is without aortotomy and relatively straightforward to perform (2). In identifying the correct location for the new ostia, we determined that filling the heart with cardioplegia adequately helps visualize the new ostia takeoff location and anticipate proper length to alleviate the vessel stretching or kinking.
Looking back, we see reimplantation was avoided in patients with intramural segments because the technique was unable to address intramurality. However, recent data indicate intramurality rather than the ostial location generates the potential ischemic effects of AAOCA (4-6). Yet there remain opposing viewpoints that suggest intramural length does not correlate with symptoms (7). Even so, reimplantation success rates of AAOCA with intramural segments have been equivalent to intramural unroofing techniques (8). Additionally, not addressing the intramural component does not appear to increase the likelihood of postoperative neo-ostial stenosis as previously thought (9). Nonetheless, unroofing has been the operation of choice, and while it has been successful (10,11), recent data emerging indicate unroofing may have more complications than we understand. Specifically, AAOCA unroofing procedures often require takedown and recoupling of the intracoronary commissure. This exposure, along with aortotomy, increases the risk of aortic valve complications requiring postoperative aortic valve replacement (12,13). However, routine commissural resuspension of the aortic valve may reduce postoperative valvular complications (12). More extensive repairs requiring aortotomy seen in unroofing, osteoplasty, or the large patch repairs seen in pulmonary artery translocation may be more susceptible to patch aneurysms, subject to longer aortic cross-clamp times, and extended time on cardiopulmonary bypass. In a single report of unroofing alone, the mean aortic cross-clamp time was 33 minutes (14), and a single report for reimplantation aortic cross-clamp time was 25 minutes (2). However, this may be more a surgeon-dependent factor as our average aortic cross-clamp time was 41 minutes. Furthermore, unroofing may not be the ideal approach due to the propensity of ischemic symptoms to persist post-operatively. One center reports that 46% of their 63 pediatric patients maintained or developed symptoms after unroofing intramural AAOCA (11). Additionally, a recent 2020 report from Gaillard et al. points out that some centers have reported nearly 1/3 of unroofing patients exhibit subclinical ischemic manifestations postoperatively (15). While their data is not free from ischemic complications (self-proclaimed), they purport osteoplasty or reimplantation techniques may resist ischemic complications long-term (15). Overall, given intramural AAOCA predisposition towards clinical symptoms and the potential persistence of symptoms after unroofing, it may indicate a benefit for reimplantation over unroofing. This is inconsistent with many centers' perspective that primarily use unroofing for AAOCA with intramural segments (11). We should consider that performing unroofing solely in intramural AAOCA segments may not be our only option. Reimplantation has been successful in our hands and is the operation of choice at some centers, even in those with intramural AAOCA (8,9,15). Despite previous success with unroofing, it may be time to reevaluate and possibly look to reimplantation as the gold standard approach for correcting anomalous coronary arteries.
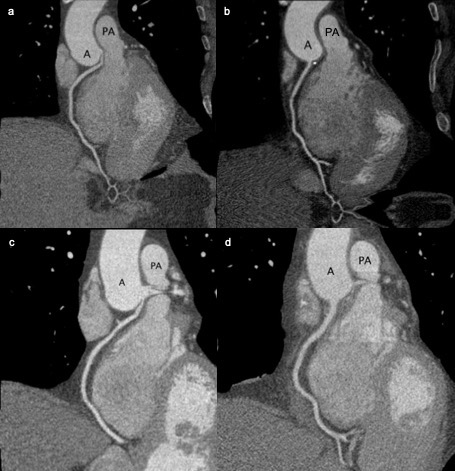
Figure 1: a. Patient 1: pre-operative CT coronary angiography that showcases the impingement of the RCA between the aorta and the pulmonary trunk. b. Patient 1: post-operative CT coronary angiography that showcases the correction of the RCA course. c. Patient 2: pre-operative CT coronary angiography that showcases the impingement of the RCA between the aorta and the pulmonary trunk. d. Patient 2: post-operative CT coronary angiography that showcases the correction of the RCA course.
References
- Carrel T. Surgical Treatment of Anomalous Aortic Origin of Coronary Arteries: The Reimplantation Technique and Its Modifications. Operative Techniques in Thoracic and Cardiovascular Surgery. 2017;21(3):178-201.
- Izumi K, Wilbring M, Stumpf J, Matschke K, Kappert U. Direct reimplantation as an alternative approach for treatment of anomalous aortic origin of the right coronary artery. Ann Thorac Surg. 2014;98(2):740-742.
- Baciewicz FA, Jr. Anomalous aortic origin of the right coronary artery. Ann Thorac Surg. 2015;99(4):1491.
- Mainwaring RD, Murphy DJ, Rogers IS, et al. Surgical Repair of 115 Patients With Anomalous Aortic Origin of a Coronary Artery From a Single Institution. World J Pediatr Congenit Heart Surg. 2016;7(3):353-359.
- Joggerst S, Monge J, Uribe C, Sherron S, Angelini P. Sudden cardiac arrest at the finish line: in coronary ectopia, the cause of ischemia is from intramural course, not ostial location. Tex Heart Inst J. 2014;41(2):212-216.
- Kaushal S, Backer CL, Popescu AR, et al. Intramural coronary length correlates with symptoms in patients with anomalous aortic origin of the coronary artery. Ann Thorac Surg. 2011;92(3):986-991; discussion 991-982.
- Balasubramanya S, Monge MC, Eltayeb OM, et al. Anomalous Aortic Origin of a Coronary Artery: Symptoms Do Not Correlate With Intramural Length or Ostial Diameter. World J Pediatr Congenit Heart Surg. 2017;8(4):445-452.
- Cubero A, Crespo A, Hamzeh G, Cortes A, Rivas D, Aramendi JI. Anomalous Origin of Right Coronary Artery From Left Coronary Sinus-13 Cases Treated With the Reimplantation Technique. World J Pediatr Congenit Heart Surg. 2017;8(3):315-320.
- Law T, Dunne B, Stamp N, Ho KM, Andrews D. Surgical Results and Outcomes After Reimplantation for the Management of Anomalous Aortic Origin of the Right Coronary Artery. Ann Thorac Surg. 2016;102(1):192-198.
- Padalino MA, Franchetti N, Sarris GE, et al. Anomalous aortic origin of coronary arteries: Early results on clinical management from an international multicenter study. Int J Cardiol. 2019;291:189-193.
- Sachdeva S, Frommelt MA, Mitchell ME, Tweddell JS, Frommelt PC. Surgical unroofing of intramural anomalous aortic origin of a coronary artery in pediatric patients: Single-center perspective. J Thorac Cardiovasc Surg. 2018;155(4):1760-1768.
- Yerebakan C, Ozturk M, Mota L, et al. Complete unroofing of the intramural coronary artery for anomalous aortic origin of a coronary artery: The role of commissural resuspension? J Thorac Cardiovasc Surg. 2019;158(1):208-217 e202.
- Romp RL, Herlong JR, Landolfo CK, et al. Outcome of unroofing procedure for repair of anomalous aortic origin of left or right coronary artery. Ann Thorac Surg. 2003;76(2):589-595; discussion 595-586.
- Sharma V, Burkhart HM, Dearani JA, et al. Surgical unroofing of anomalous aortic origin of a coronary artery: a single-center experience. Ann Thorac Surg. 2014;98(3):941-945.
- Gaillard M, Pontailler M, Danial P, et al. Anomalous aortic origin of coronary arteries: an alternative to the unroofing strategy. Eur J Cardiothorac Surg. 2020;58(5):975-982.
Diclaimer
The information and views presented on CTSNet.org represent the views of the authors and contributors of the material and not of CTSNet. Please review our full disclaimer page here.





Comments