ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Emergent Repair of the Ascending Aorta in a Patient With Loeys-Dietz Syndrome: A Mandate for Early Aggressive Management
Abstract
The authors present a case of acute aortic dissection in a patient with Loeys-Dietz Syndrome type II, complicated by multiple blood antibodies. A 68-year-old Caucasian woman presented with a sudden onset of severe, crushing chest pain. Her work-up revealed an acute intramural hematoma in the aortic root, and aortic rupture could not be ruled out. The emergency surgical repair was complicated by unavailability of matched blood, but due to the urgency of the patient’s condition, it was decided to proceed with the operation. She was taken emergently to the operating room for replacement of her ascending aorta with re-suspension of the aortic valve. Her postoperative course was complicated by a transfusion reaction.
Introduction
Loeys-Dietz Syndrome (LDS) is a rare autosomal dominant connective tissue disorder that results from mutations in the genes TGFBR1 and TGFBR2, which code for transforming growth factor β receptors 1 and 2, respectively, as well as TGFB2 and SMAD3 (1, 2). The syndrome is characterized by a classic triad of widely spaced eyes, cleft palate of bifid uvula, and vascular aneurysms, tortuosity, or dissections (2).
Genetically predisposed patients tend to develop aortic dissections at an earlier age when compared to patients without the genetic conditions (3). According to the International Registry of Acute Aortic Dissection (IRAD), after onset of type A aortic dissections, medical treatment alone leads to an hourly growth in mortality rates, reaching as high as ~22% in the first 24 hours (4).
Case Report
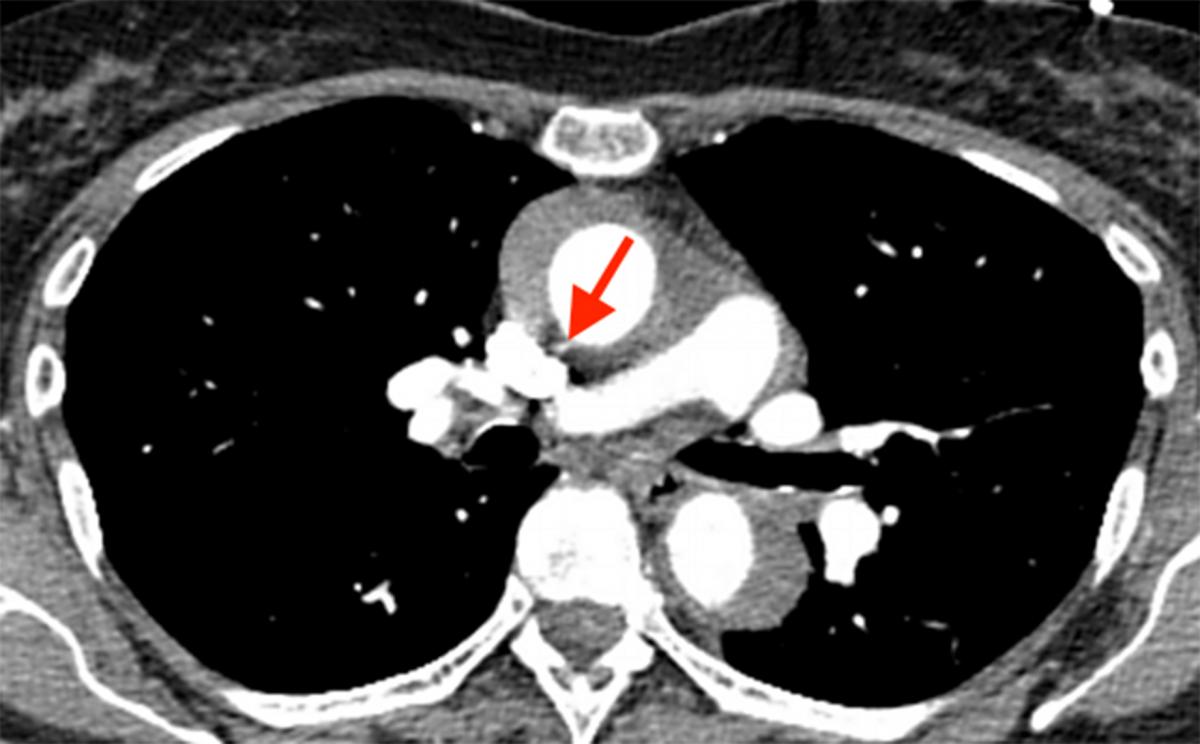
Figure 1: Computed tomography angiography scan
A 68-year-old Caucasian woman developed a sudden onset of severe crushing chest pain while shopping. She had a known history of hypertension and Loeys-Dietz Syndrome type II. She was also followed as an outpatient for a dilated ascending aorta with serial CT scans. The last known dimension was 4.1 cm. At her acute presentation, a CTA done at an outside hospital showed intramural hematoma extending from the aortic root to the suprarenal aorta. There was no obvious evidence of a false lumen.
Upon transfer to the authors’ hospital, the patient was initially monitored in the intensive care unit where her pain was well controlled and she remained hemodynamically stable. Transthoracic echocardiography demonstrated a dilated proximal aortic root with effacement of the sinotubular junction and trace aortic insufficiency, as well as a hematoma of the anterior distal aortic arch. She was then taken emergently to the operating room for repair of the ascending aorta, since intramural hematoma is known to have similar clinical consequences to aortic dissection (3). As usual, a blood specimen was sent for type and cross-match. The patient had a history of four blood antibodies and thus a cross-matched blood was not available at the time of surgery.
At the time of surgery, transesophageal echocardiogram (TEE) confirmed significant intramural hematoma in the ascending aorta, as well a raised suspicion for aortic rupture (Fig. 1). Upon opening the pericardium, the ascending aortic pathology was obvious with the typical discolored appearance. Cardiopulmonary bypass was established via the left femoral artery and right atrium.
Hypothermic circulatory arrest was instituted. The aorta was then opened and the arch was directly examined. No evidence of a tear in the arch itself was seen, but the ascending aorta had an intramural hematoma that contained a fresh clot. The aortic valve appeared to be competent. The ascending aorta was excised and an open distal anastomosis was then performed to a 26 mm Dacron graft. Antegrade flow was established by cannulation of the graft, and cardiopulmonary bypass re-established with re-warming to normothermia. The aorta was transected at the sinotubular junction and the non- coronary sinus was repaired using BioGlue and the standard suture technique. The right and left sinus portions were not affected. The proximal transected aorta was prepared and a graft anastomosis was performed. The procedure was completed in the usual manner.
The patient’s preoperative hemoglobin was 12.0 g/dl. Prior to the termination of the cardiopulmonary bypass, the measured Hgb was 5.1 g/dl. After re-transfusion of the salvaged blood, the Hgb rose to 7.4 g/dl. Because of the patient’s antibodies, no PRBC were transfused. After Heparin reversal, 1 unit of platelets, 10 units of cryoprecipitate, and factor VII were administered.
Two hours into the patient’s postoperative course, her hemoglobin remained at 7.4 g/dl, chest tube output continued to be moderate, she exhibited metabolic acidosis, and her vitals were stable. A decision was made by the ICU staff to transfuse the patient with type-specific unmatched PRBCs. Immediately upon initiation of transfusion the patient experienced acute hypotension and what appeared to be a transfusion reaction. Further transfusions were held until crossmatched blood was available. Her hemoglobin continued to drop and six hours later, at the level of 2.5 g/dl, cross-matched blood was finally made available. She was given four units of PRBC and her hemoglobin stabilized at 10.5 g/dl. The remainder of the patient’s postoperative course was remarkable only for transient renal insufficiency and her distal aorta was managed conservatively.
Comment
A familial incidence of aortic disorders has been followed in this patient’s family since 1977, with a mutation of C1459T in the TGFBR1 gene and multiple relatives affected by aortic aneurysms or dissections (5). They were classified as LDS after the process was described in 2005 by Loeys, Dietz, and colleagues (1).
Although LDS represents a clinical continuum with genetic and phenotypic similarity to Marfan’s syndrome, LDS is noted to affect more organ systems (2). It is also known to be a more aggressive process with respect to acute aortic pathology (1, 6).
Once the diagnosis of LDS is established, the patient is carefully managed with frequent cardiovascular imaging surveillance, beta-blockers, and often early surgical intervention (2). Surgical intervention should even be considered in children when the maximum diameter of the aorta exceeds the 99th percentile and the aortic annulus exceeds 1.8-2.0 cm (2, 7, and 8). In adults, surgical repair should be recommended once the aortic root diameter reaches 4 cm, or the diameter of the descending thoracic aorta reaches 5 cm. As always, rapid expansion of any of the above dimensions at a rate of over 0.5 cm a year warrants surgical repair, as well (2, 7, and 8).
The patient’s last known ascending aortic dimension was 4.1 cm. Had this patient been referred for an elective aortic replacement at that time, she may not have incurred the risks of emergent operation, which in this case included severe anemia leading to renal insufficiency.
References
- Loeys B, Chen J, Neptune E, et al. A syndrome of altered cardiovascular, craniofacial, neurocognitive and skeletal development caused by mutations in TGFBR1 and TGFBR2. Nat Genet 2005;37:275–81.
- Loeys BL, Dietz HC. Loeys-Dietz Syndrome. 2008 Feb 28 [Updated 2013 Jul 11]. In: Pagon RA, Adam MP, Ardinger HH, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2015.
- Ziganshin A and Elefteriades A. Treatment of Thoracic Aortic Aneurysm: Role of Earlier Intervention. Semin Thoracic Surg 27:135–143
- Hagan P, Nienaber C, Isselbacher E, et al. The International Registry of Acute Aortic Dissection (IRAD). AMA, February 16, 2000—Vol 283, No. 7
- Nicod P, Bloor C, et al. Familial Aortic Dissecting Aneurysm. JACC Vol. 13, No. 4 March 15. 1989:811-9
- Loeys BL, Schwarze U, Holm T, et al. Aneurysm syndromes caused by mutations in the TGF-b receptor. N Engl J Med 2006;355:788–98.
- Hiratzka L, Bakris G, et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM Guidelines for the Diagnosis and Management of Patients With Thoracic Aortic Disease. JACC Vol. 55, No. 14, 2010
- Williams J, Hanna J, et al. Adult Surgical Experience With Loeys-Dietz Syndrome. Ann Thorac Surg 2015;99:1275–81




