ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Preventing Recurrence: Optimal Surgical and Medical Management of Catamenial Pneumothorax
M. Clark, James; Brown, Lisa M.; Holmes, Sarah; Cooke, David T.; David, Elizabeth A. (2017): Preventing Recurrence: Optimal Surgical and Medical Management of Catamenial Pneumothorax.
CTSNet, Inc. https://doi.org/10.25373/ctsnet.5557981
Retrieved: 20:58, Nov 06, 2017 (GMT)
In this video, the authors demonstrate the use of video-assisted thoracoscopic surgery (VATS) in the treatment of catamenial pneumothorax in a patient with significant diaphragmatic eventration.
Thoracic endometriosis remains the most common extrapelvic location for endometrial implants, with catamenial pneumothorax occurring in 75% of thoracic endometriosis patients.1 These patients have recurrent pneumothoraxes, defined as occurring cyclically within 72 hours of the onset of menses. These pneumothoraxes may not occur or become symptomatic with every monthly cycle, but represent a significant cause for recurrent pneumothoraxes in menstruation-aged women. Though uncommon overall, catamenial pneumothoraxes constitute 3% to 6% of all spontaneous pneumothoraxes. The symptoms are similar to traditional pneumothoraxes, with chest or scapular pain and dyspnea.
Over 90% of all catamenial pneumothoraxes are right-sided.2 The exact reason for this is unclear, but one of the proposed etiologies for catamenial pneumothoraxes is that inherent or acquired microperforations or defects in the diaphragm allow for translocation of subdiaphragmatic endometrial implants into the intrathoracic space. Deposits on the right hemidiaphragm might be propelled into the right hemithorax by a piston-like effect of the inferior solid liver, imparting elevated intraabdominal pressure spikes against the already weakened right hemidiaphragm. While one-third of catamenial pneumothorax patients are found to have macroscopic diaphragmatic defects at the time of surgery, persistent diaphragmatic lesions are thought to represent a major reason for recurrence after surgery.3 Pneumothorax is then thought to develop due to transdiaphragmatic endometrial implants on the visceral pleura, which undergoes the hormonally regulated desquamation of the menstrual cycle, leading to alveolar injury and air leak.2 Additionally, there is evidence in animal models that endometrial implants can embolize into the pulmonary vasculature, where their apoptotic cycle can similarly cause alveolar damage.3
Historically, catamenial pneumothoraxes have had a high recurrence rate after surgery, ranging from 30% to 60%.5-8 Introduction of diaphragmatic mesh for repair of visible diaphragm defects or eventrations, as well as empiric use for occult diaphragm lesions, have helped reduce this rate to close to zero in multiple case series.7-8 Additionally, the use of perioperative hormonal therapy, specifically gonadotropin releasing hormone agonists, for six to 12 months postoperatively, have helped reduce recurrence even further and remain the standard care for many practitioners.8-9
The authors describe the successful surgical and medical management of a young woman with recurrent catamenial pneumothoraxes. A 34-year-old woman was referred to thoracic surgery clinic for recurrent episodes of dyspnea. She stopped taking oral contraceptive pills at the age of 30, shortly after which she began having recurrent episodes of dyspnea, cough, and dysmenorrhea. She underwent a diagnostic laparoscopy with evidence of pelvic endometriosis. She was found to have a right pneumothorax and had a chest tube placed. The pneumothorax persisted and she underwent a VATS blebectomy and decortication at an outside hospital.
Several months later, she began in vitro fertilization therapy. She developed similar right-sided symptoms, was found to have a right pneumothorax, and underwent pigtail thoracostomy. At this time, she was started on Depo-Provera therapy due to the correlation of her symptoms with hormonal treatments and her natural menstrual cycle. Seven months later she experienced a stillbirth and shortly thereafter was found to have another right-sided pneumothorax. This was again treated with pigtail thoracostomy, and she was referred to our thoracic surgery clinic. She is an otherwise healthy woman with no smoking history.
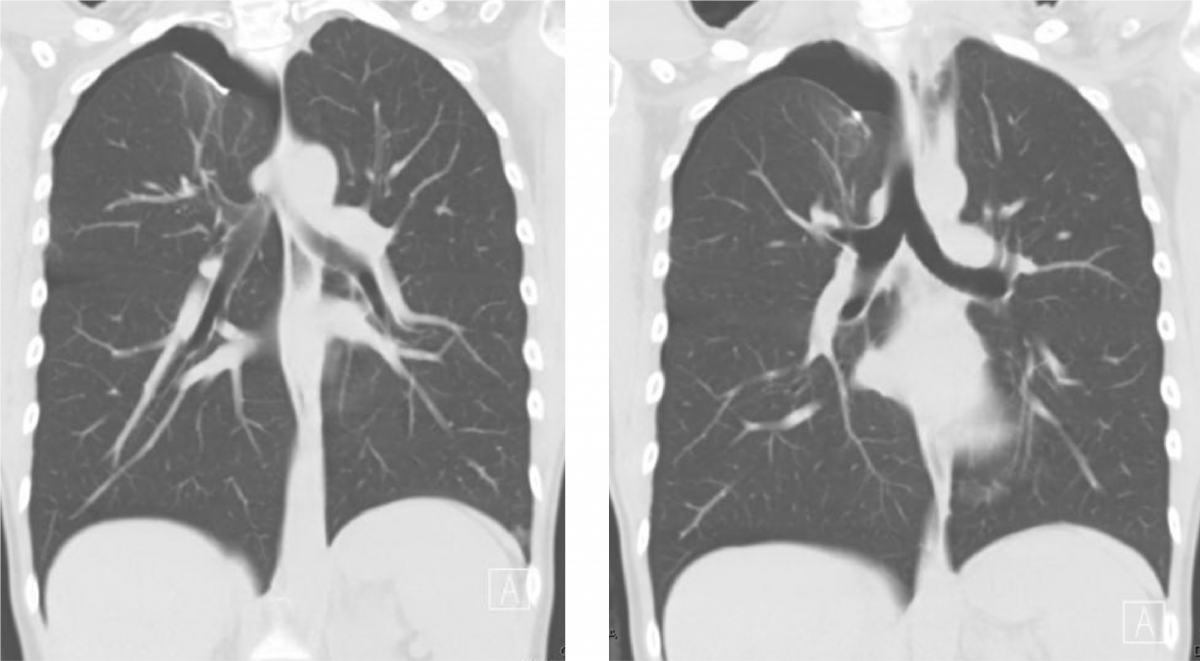
Figure 1. Selected images of the patient’s preoperative computed tomography imaging showing her right apical and basilar pneumothoraxes and pigtail thoracostomy tube.
Figure 1 shows representative images of the patient’s preoperative computed tomography (CT) scan indicating moderate right-sided apical and basilar pneumothoraxes, as well as the pigtail thoracostomy and the prior blebectomy staple line. We offered her a VATS blebectomy, apical pleurectomy, mechanical pleurodesis, and diaphragmatic repair given her persistent air leak with the tube thoracostomy, her frustration with her recurrent symptoms, and her desire to safely become pregnant again in the future.
In the operating room, after intubation and a normal bronchoscopy, she was placed in a left lateral position. A 2 cm port was placed in the posterior axillary line at the 8th intercostal space, through which a 10 mm thoracoscope was passed.
On examination, there were significant adhesions between the right upper lobe and the chest wall along the major fissure, likely from her prior VATS. An additional port was placed at the anterior axillary line in the 4th intercostal space. There was an endometrial implant on the parietal pleura that was removed and sent for pathology. A ring forceps was used in conjunction with Bovie electrocautery to take down the adhesions in order to prevent recurrence with a loculated pneumothorax.
Multiple blebs were observed on the lateral surface of the middle lobe and the apex of the upper lobe. These were removed with wide resection using an endoscopic stapler. The apical parietal pleura was scored with Bovie electrocautery. The pleurectomy was performed by bluntly dissecting the apical parietal pleura away from the chest wall. A mechanical pleurodesis was performed with a folded Bovie scratch pad on a ring forceps.
Visualization of the right hemidiaphragm demonstrated a significant diaphragmatic eventration. This was repaired with placement of a 15 x 15 cm Vicryl mesh, positioned in order to cover all macroscopic diaphragmatic defects. The mesh was fixed in place with multiple figure-eight #1 Vicryl stitches. Other microscopic occult defects were addressed with coating the mesh surface with 10 g of Tisseal fibrant sealant.
Doxycycline (500 mg) was injected into the pleural space as a chemical pleurodesis. Two 19 Fr Blake chest tubes were placed
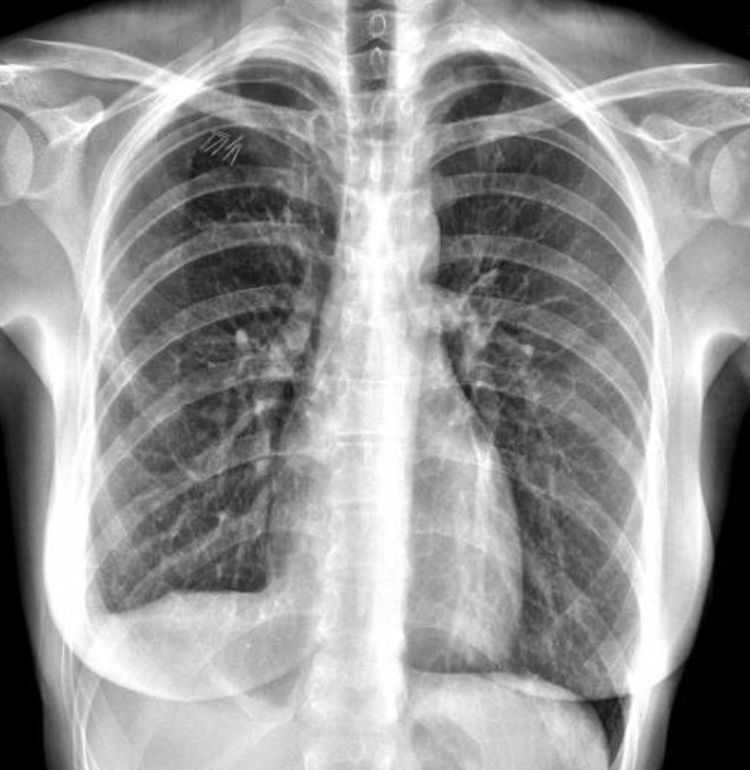
Figure 2. The patient’s 14 month postoperative chest x-ray showing no recurrence of her pneumothorax.
The patient had an uncomplicated hospital course and was discharged on postoperative day three after her chest tubes were removed. Lupron was administered within one week of discharge. She remains on Lupron Depot intramuscular shots for one year postoperation to reduce the chance of recurrent catamenial pneumothorax. She has been without recurrent pneumothorax as seen in the chest x-ray in Figure 2 at her last follow-up 14 months after surgery. Note the diaphragm patch in place on the right hemidiaphragm.
In conclusion, catamenial pneumothorax is a morbid condition that can be effectively managed with appropriate surgical and medical therapy. Surgical management should include blebectomy, apical pleurectomy, pleurodesis, and, as recently demonstrated, appropriate repair of macroscopic and coverage of microscopic diaphragm defects. Medical management should include six to 12 months of hormone suppression with a gonadotropin releasing hormone agonist. Isolated surgical repair without appropriate medical management is likely to result in high rates of recurrence.
References
- Bagan P, Berna P, Assouad J, Hupertan V, Le Pimpec Barthes F, Riquet M. Value of cancer antigen 125 for diagnosis of pleural endometriosis in females with recurrent pneumothorax. Eur Respir J. 2008;31(1):140-142.
- Legras A, Mansuet-Lupo A, Rousset-Jablonski C, et al. Pneumothorax in women of child-bearing age: an update classification based on clinical and pathologic findings. Chest. 2014;145(2):354-360.
- Roberts LM, Redan J, Reich H. Extraperitoneal Endometriosis With Catamenial Pneumothoraces: A Review of the Literature. JSLS. 2003;7(4):371-375.
- Korom S, Canyurt H, Missbach A, et al. Catamenial pneumothorax revisited: clinical approach and systematic review of the literature. J Thorac Cardiovasc Surg. 2004;128(4):502-508.
- Alifano M, Jablonski C, Kadiri H, et al. Catamenial and noncatamenial, endometriosis-related or nonendometriosis-related pneumothorax referred for surgery. Am J Respir Crit Care Med. 2007;176(10):1048-1053.
- Marshall MB, Ahmed Z, Kucharczuk JC, Kaiser LR, Shrager JB. Catamenial pneumothorax: optimal hormonal and surgical management. Eur J Cardiothorac Surg. 2005;27(4):662-666.
- Bagan P, Le Pimpec Barthes F, Assouad J, Souilamas R, Riquet M. Catamenial pneumothorax: retrospective study of surgical treatment. Ann Thorac Surg. 2003;75(2):378-381.
- Ciriaco P, Muriana P, Bandiera A, Carretta A, Negri G, Candiani M, Zannini P. Multidisciplinary Approach to Catamenial and Endometriosis-Related Pneumothorax. Southern Thoracic Surgical Association: 63rd Annual Meeting Abstract Book. Naples, FL, 2017 Jan, 359.
- Attaran S, Bille A, Karenovics W, Lang-Lazdunski L. Videothoracoscopic repair of diaphragm and pleurectomy/abrasion in patients with catamenial pneumothorax: a 9-year experience. Chest. 2013;143(4):1066-1069.




