ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Trans-Aortic Transcatheter Aortic Valve Replacement with Edwards Sapien-Ascendra 3
Dr. Mark Russo and Dr. Paolo Tartara are proctors for Edwards Lifesciences. Dr. Tartara is also a full-time employee of Edwards Lifesciences.
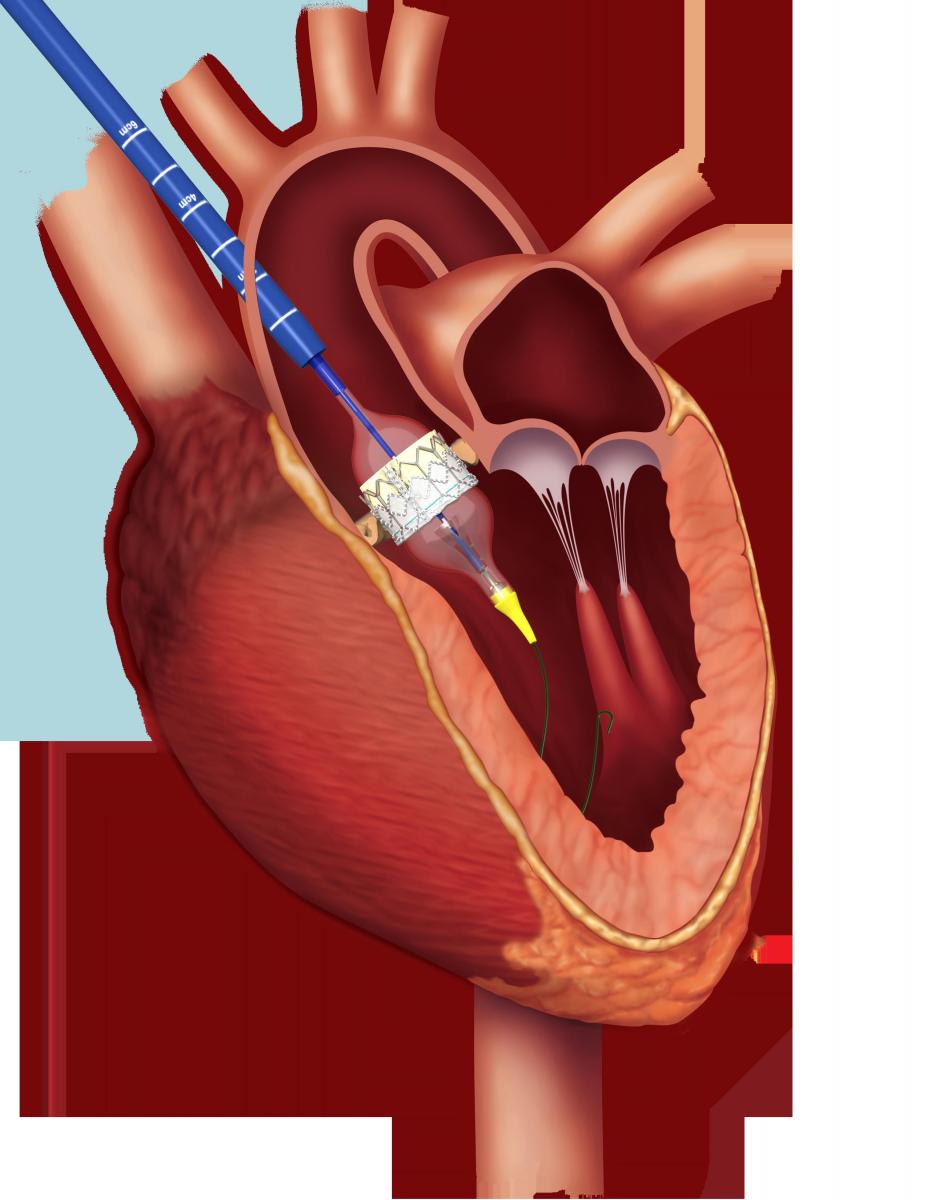
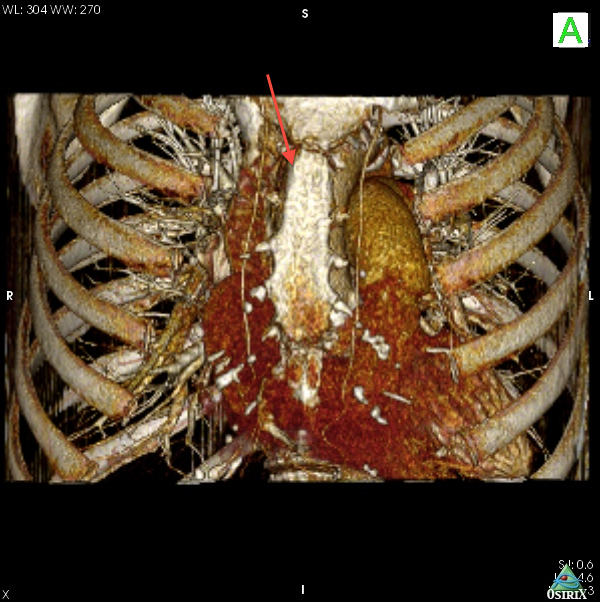
Transcatheter aortic valve replacement (TAVR) is indicated for patients with severe, symptomatic aortic stenosis, who are deemed to be unsuitable candidates for high-risk surgical aortic valve replacement (SAVR). For patients who have insufficient iliofemoral access, the trans-femoral route is the preferred approach for the procedure. However, in patients with inherently small arteries and/or severe peripheral vascular disease, alternative trans-aortic and trans-apical access routes are used. This article describes a standardized and reproducible trans-aortic TAVR approach with the Edwards Sapien valve and Ascendra 3 delivery system (Fig. 1,2).
Fig. 2: The Ascendra 3 delivery system has a nose cone (yellow) to facilitate retrograde crossing of the native aortic valve.
Introduction
Transcatheter aortic valve replacement (TAVR) outcomes have shown promising results in recent studies. Survival is equivalent between high-risk patients who receive surgical aortic valve replacement (SAVR) and TAVR [1], and survival is significantly improved in inoperable patients who receive TAVR over medical therapy [2]. Recently, the United States Food and Drug Administration (FDA) approved “alternative access approaches,” for TAVR (three [2], based on the increasing body of evidence supporting its use in appropriately selected patients [3,4]).
Patient Selection
A trans-aortic approach is preferred over a trans-apical approach in patients who are have compromised trans-femoral access in the following clinical settings:
- Severe pulmonary disease compromising the pleural space
- Disease of the descending and abdominal aorta, including:
- Severe calcification/atheroma of the aortic arch
- History of aortic occlusion, aortic aneurysm, or aortic dissection - Other vascular disease leaving no access for bailout cardiopulmonary bypass (e.g., both iliofemoral and subclavian arteries are not suitable for cannulation)
- Left ventricular functional or anatomical abnormalities, including:
- Severe systolic or diastolic dysfunction
- Small ventricular cavity, with severely hypertrophic myocardium
- Thin left ventricular wall, or severe dilated cardiomyopathy
- Extremely frail myocardial tissues
- Chronic steroid therapy
Contraindications for a Trans-Aortic Approach
- Calcified or atheromatous ascending aorta
- Vein grafts with very high origin
- Cardiac anatomic variations, including:
- Grafts close to the sternum, placing patient at risk for sternotomy
- Variations preventing a good coaxial prosthesis deployment (e.g., pectus excavatum)
- Absence of pathological processes (e.g., calcification, atheroma, thrombi, dissection flap) or evidence of previous surgery (e.g., pledget)
- At least 1 cm distal to coronary venous grafts or foreign bodies (e.g., Teflon felt from previous aortic cannulation)
- At least 5.5 cm distant from the aortic annulus to allow sufficient room for:
- the tip of sheath to enter the aorta (2 cm)
- full balloon expansion (3 cm, as half of the balloon will be staying in the aortic root). See Figures 1 and 3. - Aligned sheath placement with aortic annulus in order to achieve a coaxial prosthesis deployment.

Fig. 3 The delivery system balloon is 5.5 cm long, from the proximal to the distal end. The picture shows the Ascendra 1 delivery system, without the yellow nose cone.
2. Determine the preferred surgical access needed to gain access to the puncture site.
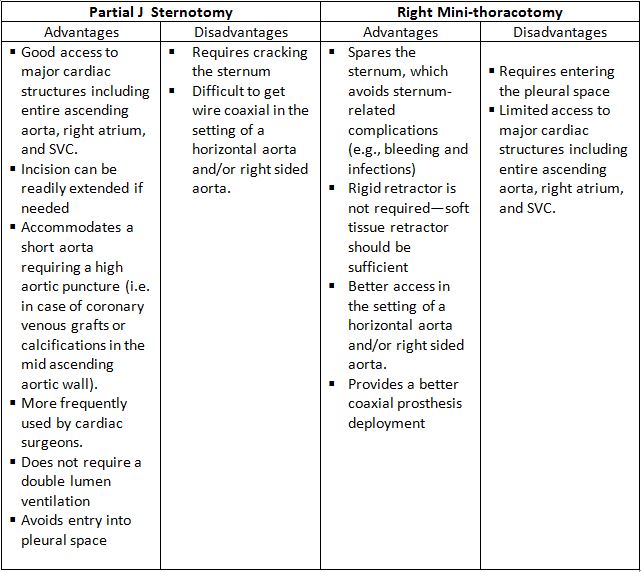
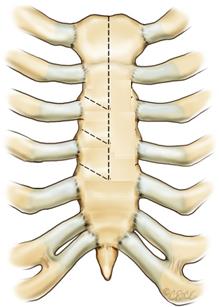
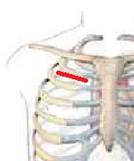
There are two minimally invasive surgical approaches: partial J-sternotomy and right minithoracotomy (Figure 4).
Table 1. Advantages and Disadvantages of the Partial J Sternotomy and Right Mini-thoracotomy
Fig. 4 Ministernotomy (right) and minithoracothomy (left).
How to Conduct the CT Analysis
- Evaluate the ascending aorta between the mid portion and the origin of the innominate artery. Evaluation should assess the right anterolateral aspect of the aorta for aortic wall disease and venous grafts.
- Measure the distance between the aorta and the chest wall. A deep aorta (> 6 cm from the chest wall) reduces the ease of access to the aorta and may require a larger incision:
- The hemisternotomy incision may be extended to the 3rd or 4th intercostal space
- In the case of minithoracotomy, the rib above the incision may be cut at the cartilage, then reapproximated. - If the axial CT views demonstrate the aorta is central, behind the sternum, or left-sided, an upper ministernotomy is recommended. If the axial CT views demonstrate that the aorta is on the right of the sternum and/or horizontal (angle >40-45°), a minithoracotomy is suggested, unless other specific anatomical or clinical contraindications exist.
- These images provide information regarding the angle of the aortic annulus plane with respect to the horizontal plane. If the horizontal aorta angle is >40-45°, a minithoracotomy usually provides the ideal coaxial access.
- If the aorta is vertical, a ministernotomy is better. The aortic annulus angle plane can be measured in the coronal CT views. The coronal views are also used to measure the distance between the aortic annulus and the proposed aortic puncture site.
- If the horizontal aorta angle is >75°, a trans-apical approach may be preferred.
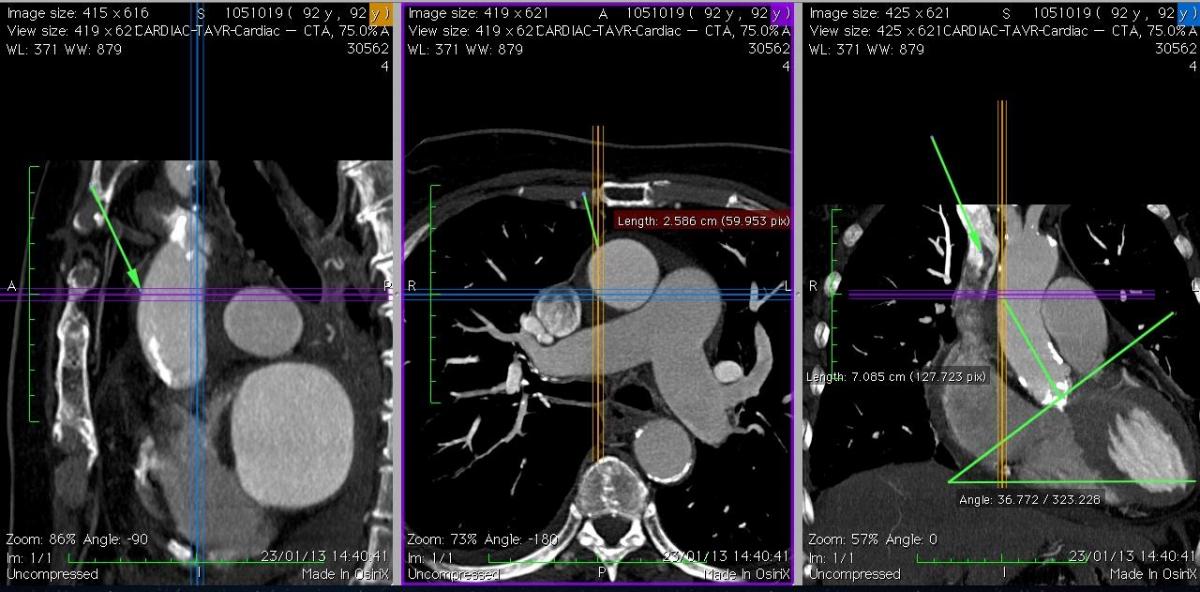
Fig. 5A: Case 1. The coronal view (right picture) shows a “vertical” aorta, and the ideal coaxial direction of the delivery system, with a suitable puncture site at 7 cm. The axial view (middle) shows that the ascending aorta is central (behind the sternum) and free of calcification at the level of the projected puncture site.
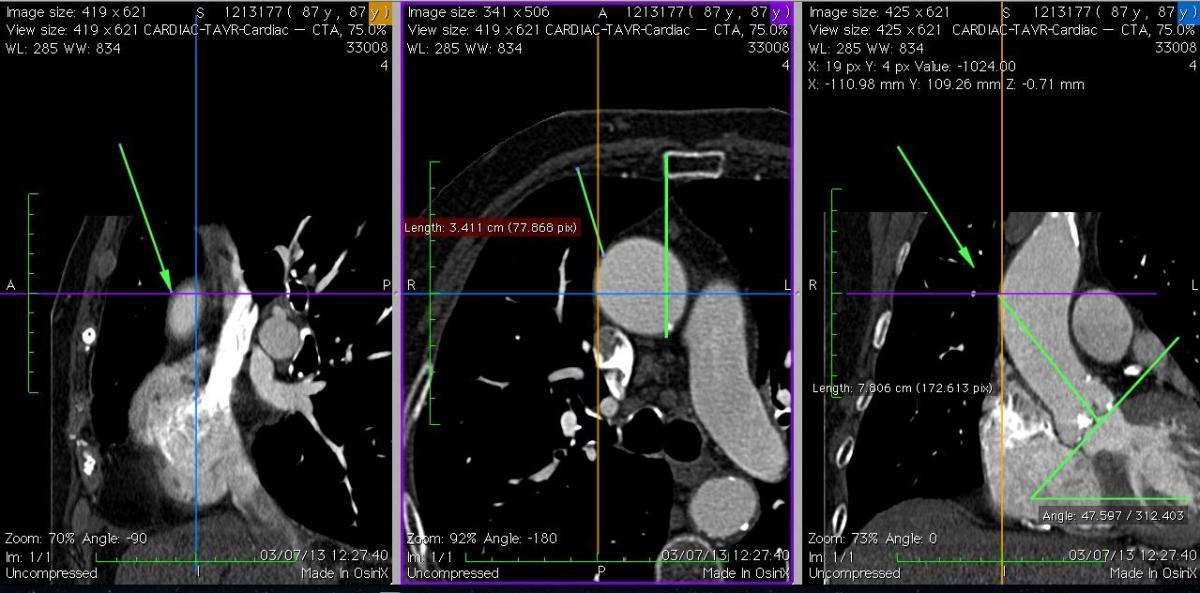
Fig. 6A: Case 2. In comparison to Case 1, the ascending aorta is more horizontal, and right-sided relative to the sternum. It was possible in this case to find a suitable puncture site at about 8 cm from the aortic annulus.
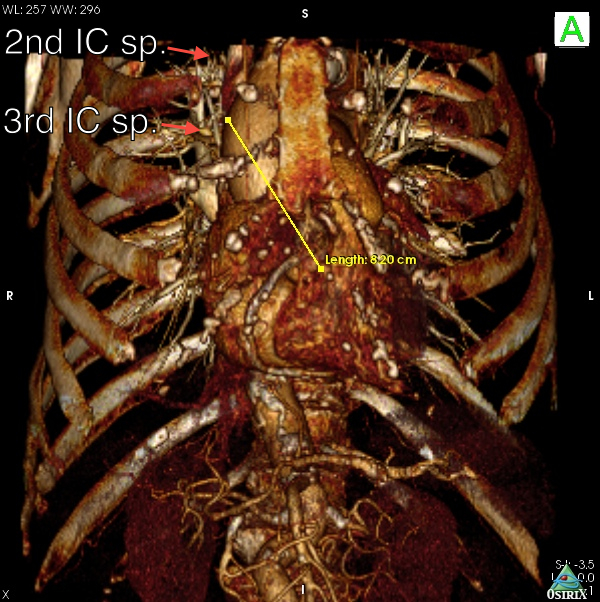
Fig. 6B: Case 2. The 3D volume rendering images revealed the right-sided and horizontal anatomy of the ascending aorta; this suggested that the optimal incision was a right upper mini-thoracotomy approach through the 2nd intercostal space.
4. Multiplanar Reconstruction
Multiplanar reconstruction CT analysis provides a very comprehensive analysis to help determine the ideal puncture location. If multiplanar reconstruction analysis technology is not available, a 3D volume rendering reconstruction analysis is recommended, in addition to the standard axial view analyses.
Operative Procedure
Room Setup
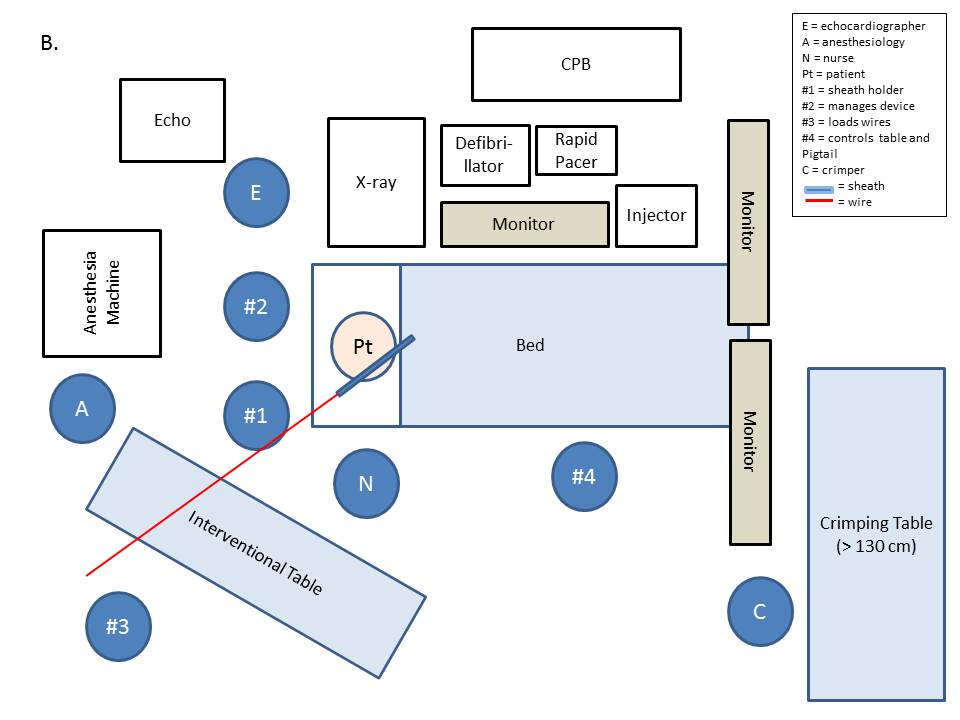
Typically a minimum of 3 operators are required to (1) manage the sheath, (2) manipulate the catheters, and (3) load and support the catheters and inflate the valve. A fourth operator may be needed to control the table and the Pigtail. Generally, the echocardiographer is to the left of the table, and the anesthesia equipment is moved back (Fig. 7A). During the procedure, the anesthesiologist has restricted access to the patient; therefore the operators must take care not to the disrupt anesthesia equipment. Long anesthesia lines facilitate these configurations.
Depending on the size and configuration of the room, the team may choose to have the distal end of the wires toward the foot of the bed (Fig. 7A); in this configuration, longer wires (> 180 cm) are needed to allow for gentle bends in the wire. Alternatively, the team may choose to have wires go straight behind the sheath (Fig. 7B); in this configuration, an additional table is needed to the right side of the patient. This setup is recommended if the room is large enough, as it allows easier handling of the wires and catheters. Figure (7C) is the preferred arrangement for a minthoracotomy. The “implanter” who manages the device and positions the valve is located in the middle, between the "holder” and the "inflater." The interventional table is at the immediate right side of the operating table. Therefore the delivery system may be supported by the table.
Fig. 7: As described above, the team may choose various approaches based on the size and configuration of the room.
Patient Preparation
Bi-axillary defibrillator pads should be used. When doing a minithoracotomy, using a double lumen tube should be considered. Peripheral access should be set up as follows:
- 1 arterial femoral access for the Pigtail
- 2 venous femoral access points
- 1 for the endocavitary pacemaker
- 1 as a bailout for venous femoral cannulation
In the unlikely situation that cardiopulmonary bypass support is required, aortic cannulation is recommended, either through the Ascendra 3 sheath or through a different aortic cannulation site. If the access to the right atrium is limited, the femoral vein is preferred.
An incision is made in the 2nd right intercostal space. Either the 2nd or the 3rd rib may be divided at the cartilage in order to improve access if needed. The subcutaneous and deep tissue is divided, and the pleural sac is opened. If exposure is limited, division of the internal mammary artery and/or disarticulation of a rib may provide better exposure. A soft tissue retractor is then inserted. Multiple pericardial stay sutures are placed in order to enhance the access to the aorta and avoid lung interference. Typically a rib spreader is not required, and its use should be avoided whenever possible, as it is associated with significant post-procedural pain.
Procedural Steps
- The aorta is punctured in the middle of the purse string with an 18 gauge needle.
- A short soft wire is passed in a retrograde fashion into the ascending aorta.
- The needle is removed and a 6 Fr sheath 17-25 cm long (with radiopaque tip) is inserted over the wire.
- The short wire is exchanged for a long soft straight wire (standard, caliber 0.035).
- The valve is crossed with the straight wire. It is usually easy to cross the valve by pointing the 6F sheath toward the aortic valve, without using a catheter. If this is not the case, a 5F multipurpose catheter may help to cross the valve.
- The 6F sheath is advanced into the LV outflow tract (2-3 cm). Then a Pigtail catheter is introduced into the LV apex and exchanged for an Amplatz extrastiff wire with a tight loop (Figure 8).
- Placement of the wire at the apex is confirmed by echo and fluoroscopy (RAO view); the wire should not be entangled in the mitral valve apparatus.
- The 6F sheath (and any catheter) is exchanged over the stiff wire with Ascendra 3 to a depth of 2 cm into the aorta.
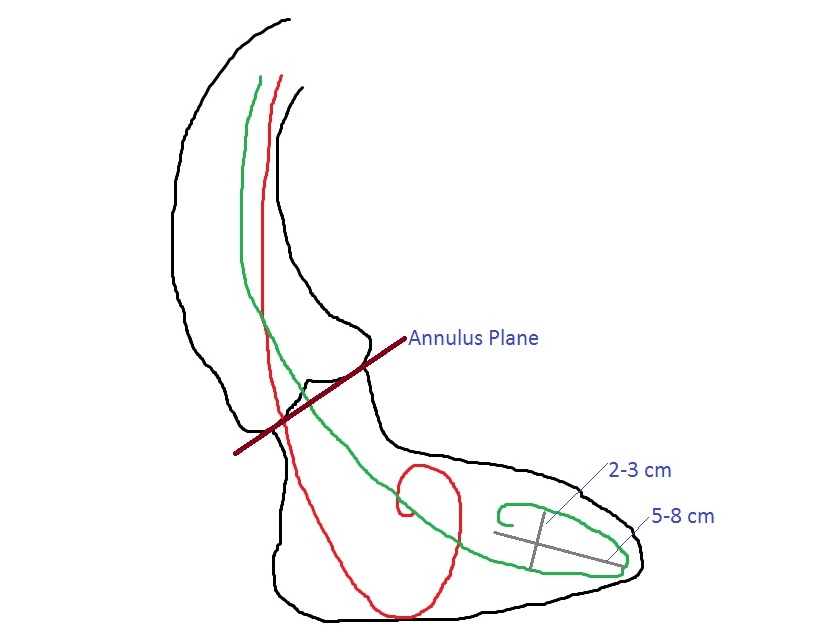
Fig. 8: The Amplatz stiff wire loop has a tight bend (green) and is positioned in the LV apex, thus allowing for a coaxial position of the delivery system and optimal prosthesis deployment. A large loop (red) prevents the wire from getting to the apex, displacing the delivery system, and leading to a canted deployment.
- The valvuloplasty balloon is passed across the annulus.
- The aortic valvuloplasty is performed under rapid ventricular pacing.
- The valvuloplasty balloon is exchanged for the Sapien Valve delivery system.
- The injector should be loaded for multiple contrast injections (3 or more).
- The delivery catheter is advanced over the wire and crosses the aortic valve with the prosthesis.
- Once the valve is intraventricular, the pusher is retracted and locked.
- The valve is positioned within the annulus.
- Pre-deployment check is conducted at three levels:
i.Valve position and coaxiality;
ii.Ascendra sheath orientation toward the annulus, and depth into the aorta;
iii.Stiff wire position into the apex. - Last-second corrections may be made to improve valve coaxiality if the positioning is poor. Corrections can be made with the following steps: 1. Modifying the sheath orientation; 2. Moving the aorta by surgeon’s finger; 3. Changing the tension on the stiff wire.
- Deployment sequence is initiated through rapid ventricular pacing starting at 180bpm (and increased as needed) in order to drop the MAP to 50 mm Hg.
- 15cc of contrast are injected into the Pigtail to confirm position of the valve within the annulus. Minor and fine positioning adjustments may be made if needed.
- The balloon is slowly inflated. If any movement is observed during the slow balloon inflation, the valve position should be corrected. Contrast is injected as needed to guide deployment.
- The balloon is deflated and the PM is turned off.
- The balloon is pulled back into the Ascendra 3 sheath.
- Echo and angiographic assessment of valve position and function are conducted. Occasionally, the valve may require an additional inflation with additional saline.
- the purse string sutures are tied to secure hemostasis
- During removal of sheath and securing purse-strings, hypotension may be induced by (a) compressing the right atrium to limit inflow to the heart or (b) a brief rapid pacing at 100-140 bpm
Tips & Pitfalls
Complications
- Cardiac arrest
- Aortic dissection
- Rupture of aortic root or annulus
- Injury to conducting system - heart block and arrhythmia
- Myocardial ischemia or infarction
- Valve migration and embolization
- Coronary ostial obstruction
- Paravalvular or central leak
- Bleeding requiring re-exploration of the chest
- Stroke
- Thrombo-embolism
- Acute renal failure
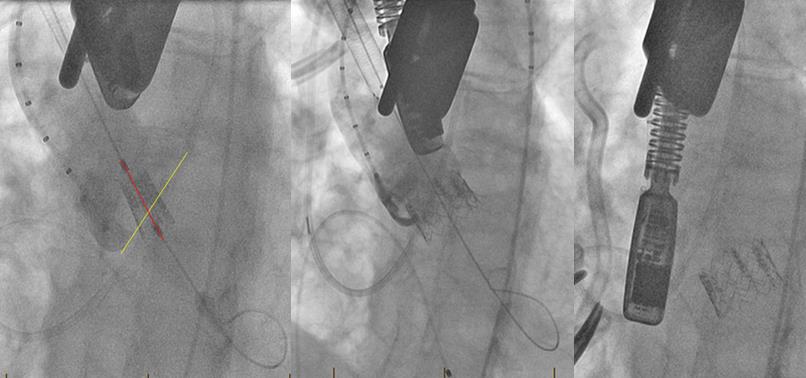
Fig. 9: Intraoperative angiograms from a case complicated by valve embolization. The wire is not coaxial with the native annulus (left). The valve is deployed in a canted fashion—50/50 on the non-coronary cusp and 10/90 on the left coronary cusp (center). Prior to removal of the sheath, the valve embolized into the ventricle (right).
Preference Card
- defibrillator pads: biaxillary
- aortic cannula
- percutanous femoral venous cannula
- center 2-stage venous cannula
- standard sternal saw
- oscillating saw
- minimally invasive sternal retractor
- hemostat
- 2 pledgeted 3-0 prolene sutures
- 2 standard 6F sheaths (Terumo Co., Somerset, NJ, USA)
- calibrated 5F Pigtail 110 cm (Cordis Corporation, Johnson Johnson, Warren, NJ, USA).
- bipolar pacer wires (Medtronic)
- standard 5F Pigtail (Cordis Corporation, Johnson Johnson, Warren, NJ, USA).
- 18 gauge needle
- short soft J-wire
- 6 Fr sheath 17-25 cm long with radiopaque tip (Terumo Co., Somerset, NJ, USA)
- long soft straight wire (standard, caliber 0.035)
- 5F Multipurpose catheter
- Amplatz Super-Stiff 0.035” x 260 cm length x 6 cm Flexible Tip Length wire (Boston Scientific Corporation, Natick, MA, USA).
- Ascendra 3 (Edwards Lifesciences, Inc., Irvine, CA, USA)
- 3 x 20 Edwards aortic valvuloplasty balloon (Edwards Lifesciences, Inc., Irvine, CA, USA)
- Sapien valve (Edwards Lifesciences, Inc., Irvine, CA, USA)
References
- Kodali SK, Williams MR, Smith CR, et al. Two-year outcomes after transcatheter or surgical aortic-valve replacement. N Engl J Med. 2012;366(18):1686-1695. doi:10.1056/NEJMoa1200384.
- Makkar RR, Fontana GP, Jilaihawi H, et al. Transcatheter aortic-valve replacement for inoperable severe aortic stenosis. N Engl J Med. 2012;366(18):1696-1704. doi:10.1056/NEJMoa1202277.
- Mack MJ, Brennan JM, Brindis R, et al. Outcomes following transcatheter aortic valve replacement in the United States. JAMA J Am Med Assoc. 2013;310(19):2069-2077. doi:10.1001/jama.2013.282043.
- Dewey TM, Bowers B, Thourani VH, et al. Transapical aortic valve replacement for severe aortic stenosis: results from the nonrandomized continued access cohort of the PARTNER trial. Ann Thorac Surg. 2013;96(6):2083-2089. doi:10.1016/j.athoracsur.2013.05.093.