ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Transaortic Valvular Implantation
Transaortic valve replacement is an alternative to the transfemoral and transapical approaches in an 83-year old male with ischemic heart disease, severe peripheral artery disease and severe symptomatic aortic stenosis.
Percutaneous aortic valve replacement is increasingly used in patients with symptomatic severe aortic stenosis in whom surgical risk is not acceptable or is very high. The most frequently used approach is transfemoral but other approaches have been used such us transapical (Edwards SAPIEN) or transaxillary-subclavian (Medtronic Corevalve®*). The transaortic approach has been proposed as an alternative to the transapical route because of proposed benefits for the patients in terms of speedier recovery and reduced morbidity.
The case concerns an 83 year old male with a history of atrial fibrillation, coronary artery disease with a bare metal stent (BMS) implanted in the right coronary artery and severe peripheral arterial disease with involvement of both femoral arteries and supra aortic trunks. At his last medical consultation, he complained of angina with mild exertion during the previous months and his last echocardiogram revealed severe aortic stenosis, moderate mitral regurgitation and severe tricuspid regurgitation, moderate left ventricular hypertrophy and mild left ventricular dysfunction. He was referred to our hospital, and a new coronary angiogram showed a new lesion in the proximal right coronary artery that was treated with two BMSs with good immediate results.
Nevertheless, he continued to experience angina that was associated with increasing dyspnea at mild effort (NYHA II-III/IV). The patient was assessed by the Heart Team of our hospital which concluded that he had an unacceptable risk for conventional aortic valve replacement (STS morbidity and mortality risk of 28%). Instead, we opted in this case for the transaortic approach because of his peripheral vascular disease and in an attempt to avoid ventricular injury.
This approach requires prior careful examination of ascending aorta by CT and by means of aortic angiograpghy. The presence of extensive calcification at the upper–anterior level of the ascending aorta is a relative contraindication to this approach. Another important consideration is the minimal distance between the annulus and the aortic junction, which should be 6 cm. This entry point is usually located just below the brachiocephalic trunk or innominate artery.
The transaortic approach was performed under general anaesthesia. A transesophageal echocardiogram revealed an aortic annulus of 20 mm and there was mild biventricular dysfunction. A pacing wire was placed in the right ventricular apex via the femoral vein and a pigtail catheter was placed through the femoral artery in the ascending aorta to monitor the arterial pressure.
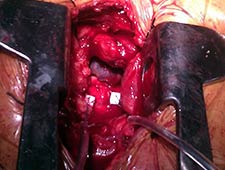
A J-shaped partial superior sternotomy allowed access to the ascending aorta after incising the pericardium. After localizing a noncalcified area over the ascending aorta, two pledgeted purse string sutures marked the incision site (Figure 1). A guide wire (Emerald 0.038, Cordis) was introduced through a 6-French catheter after puncturing the purse string, crossing the aortic valve under direct visualization with fluoroscopic guidance. This wire was replaced by an Amplatz extra-stiff wire (Cook). The Ascendra 2 Transapical Delivery System of 23 mm was introduced through its 24-French sheath, guided by the stiff crossing wire. The fluoroscopic projection is the same we use in other procedures of TAVI, left anterior oblique with some cranial angulation.
The sheath is long for the purpose of this procedure. The handling of the sheath while introducing the valve and during implantation requires good synchronization between both operators. When the position was correct, guided by angiography and transesophageal echocardiography, we proceeded with valve implantation (Edwards SAPIEN 23 mm). Rapid ventricular pacing to minimize transvalvular flow is required (Video 1, 12 sec.). As is clearly seen in the video, there was a significant superior shift upwards of the balloon-valve (not seen in our previous experience with 80 femoral and apical implantations). However, slow inflation allowed us to push the system forward slightly while not fully expanded and to finally implant the valve in an adequate position.
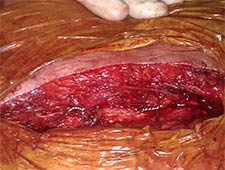
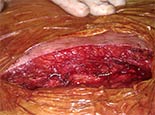
Once the valve was implanted we checked the final result with transesophageal echocardiography for any paravalvular aortic regurgitation between the right and left coronary leaflets as well as for a decrease in mitral regurgitation. An aortogram was also performed to assess the final result (Video 2, 5 sec). After sheath withdrawal, the purse string sutures were tied, requiring an additional circumferential suture for complete hemostasis. After the procedure was complete, sternal closure was performed with wire fixation (Figure 2).
During the first hours post-procedure he remained in the intensive care unit. 24 hours later he was discharged to conventional hospital ward without further incident except for an episode of bradychardia in the context of negative chronotropic drugs for atrial fibrillation. Echocardiography before discharge, 10 days later, showed mild periprosthetic aortic regurgitation and normal biventricular function.
Video 1
Video 2
The case raises some technical comments:
- The importance of pre-procedural examination of the ascending aorta, focusing on calcification and distance to the annulus.
- The sheath is not designed for this approach and is long and not very suitable. A specific sheath will be launched by the company in the near future.
- It is possible that in this approach, the short distance between aortic access and annulus and the extra-length of the sheath make the position of the valve less stable during inflation. Slow inflation and correction of potential shifts before full expansion are encouraged. The use of a marker on the sheath could help in detecting migration. In our case the shift of the sheath was unexpected. The sheath was handled by two operators like in transapical cases. We have experience in 25 transapical cases. The use of specifically designed systems could prevent this problem.
Potential benefits of this technique could be:
- Less adverse effects on respiratory function related to preservation of the diaphragm. This technique also avoids opening the left pleura and the subsequent necessity for pleural drainage.
- Management of postoperative pain is easier than using the intercostal route.
- Hemostasis is easily achieved in this location, even in fragile aortas, compared to the left ventricle.
- In patients with a low ejection fraction, scarring of the apex could adversely affect postoperative ejection fraction.
* NOTE: At the time of publication Medtronic Corevalve® is available only as an investigational device in the United States.
References
- Alegría-Barrero E, et al. Direct aortic transcatheter aortic valve implantation: severe peripheral vascular disease. Cardiovasc Revasc Med (2012)doi:10.1016/j.carrev.2011.12.004
- 2012 ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement: developed in collaboration with the American Heart Association, American Society of Echocardiography, European Association for Cardio-Thoracic Surgery, Heart Failure Society of America, Mended Hearts, Society of Cardiovascular Anesthesiologists, Society of Cardiovascular Computed Tomography, and Society for Cardiovascular Magnetic Resonance. Ann Thorac Surg. 2012 Apr;93(4):1340-95.