A 44 year old female presented with a complaint of increasing swelling in her head and upper chest over the last year. She reported increasing shortness of breath and a drowning sensation when in the supine position. Her face becomes red and tight with minimal exertion. The past medical history is significant for hypogammaglobulinemia related to IgA deficiency requiring intravenous immunoglobulin therapy for the last ten years. Additional pertinent past medical history includes placement of subcutaneous central catheters/ports on multiple occasions for delivery of immunoglobulin therapy as well as a history of venous thromboses of the lower extremities with documented bilateral pulmonary emboli requiring anticoagulation and eventually an inferior vena cava (IVC) filter.
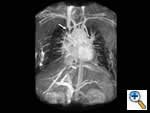 |
| Figure 1 |
On physical exam the patient had an obviously swollen head without cyanosis, but with multiple collaterals over her chest and upper neck. An MRA was obtained which demonstrated complete obstruction of the superior vena cava below the brachiocephalic junction (Figure 1). There were also significant collaterals through the intercostals and internal mammary veins, but no evidence of intrathoracic mass, adenopathy, or evidence of vein thrombosis.
Initially, the patient was seen by vascular surgery and an unsuccessful attempt at an endovascular stent placement was performed. During the stent deployment, an intraoperative venogram demonstrated normal internal jugular vein refluxing to the external jugular vein with additional refluxing to the brachiocephalic and right subclavian vein, which were both noted to be highly diseased. Furthermore, the venogram demonstrated occlusion of the superior vena cava. She was subsequently referred to thoracic surgery for evaluation of superior vena cava stenosis.
The patient was taken to the operating room where a standard median sternotomy was performed. Inspection revealed a left brachiocephalic vein that was completely obliterated and non-functioning. The SVC was normal at its junction with the right atrium, but at approximately mid level it was noted to be scarred and atretic to the level of the right internal jugular and right subclavian veins (Figures 2 and 3). Simultaneously the left saphenous vein, from inguinal canal to the ankle, was harvested endoscopically (Figure 4). Once harvested the vein was incised and opened longitudinally along its entire length. Using a standard 2-stage venous cannulae, a spiral vein graft was constructed using running 7-0 prolene suture, exchanging suture with each completed turn (Figures 5 and 6). The vein graft was left on the end of the cannulae and using a parachute technique an end-to-end anastomosis was first created in the distal SVC at the confluence of the right internal jugular and subclavian veins. The proximal anastomosis was constructed below the level of the obstruction at the entry area of the azygous vein. Following de-airing, inspection revealed the vein graft to be widely patent with no thrill or kinking (Figure 7). The patient was discharged home with coumadin with goal INR of 2.0 for 6 months. At six month follow-up, the patient reports significant improvement in her symptoms.
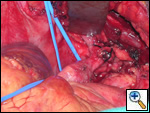 |
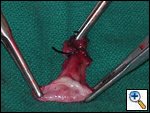 |
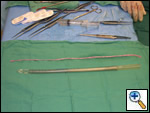 |
| Figure 2 | Figure 3 | Figure 4 |
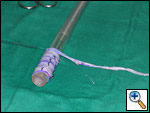 |
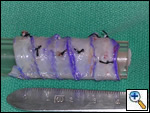 |
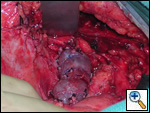 |
| Figure 5 | Figure 6 | Figure 7 |
Comments
Although malignancy is a frequent cause of SVC obstruction, benign etiologies are increasing in frequency. Iatrogenic SVC obstruction contributes to a significant proportion as evidenced by the increasing number of patients exposed to transvenous pacemaker electrodes and central venous catheters. Troubled by the lack of suitable conduit for SVC reconstruction the Canadian surgeon Ray Chiu developed and perfected the technique of spiral vein grafting in 1973 [1]. Apparently he was inspired by the common ace bandage as he thought of its “ease and adaptability in matching size when wrapping an extremity.” Chiu and colleagues ingeniously devised the new method by harvesting autologous saphenous vein, opening the vessel longitudinally, wrapping it in spiral fashion around a temporary tubular stent such as a chest tube, and then running a monofilament suture to construct the tube graft. The technique is widely versatile and has been used to replace a number of venous passages of varying sizes including the internal jugular vein, portal vein, renal vein, and common femoral vein [2]. Doty, et al, have reported on their experience of SVC bypass using the spiral vein graft and demonstrated excellent long-term patency rates (87.5% patency at a mean of 10.9 years) [3]. The use of spiral vein grafting in benign SVC obstruction is superior to either PTFE grafting or nonsurgical interventions such as PTCA or stenting [4,5]. In summary, spiral vein grafting for benign SVC obstruction demonstrates excellent long-term results with nearly 90% of patients experiencing total resolution of symptoms relating to the SVC syndrome. In addition to benign disease, spiral vein grafting may well be useful for malignant causes of SVC obstruction and in other situations where there is the need for a large caliber vein graft.
References
- Chiu CJ, Terzia J, and MacRae ML. Replacement of the superior vena cava with the spiral composite vein graft: a versatile technique. Ann Thorac Surg 1974;17:555-60.
- Chiu CJ. Spiral vein graft: a historical vignette. Canad Jour Surg 1998;41(1):8.
- Doty JR, Flores JH, and Doty DB. Superior vena cava obstruction bypass using spiral vein graft. Ann Thorac Surg 1999 67:1111-6.
- Gloviczki P, Pairolero PC, Toomey BJ, et al. Reconstruction of large veins for nonmalignant venous occlusive disease. J Vasc Surg 1992;16:750-61.
- Wisselink W, Money SR, Becker MO, et al. Comparison of operative reconstruction and percutaneous ballon dilatation for central venous obstruction. Am J Surg 1993;166:200-5.




