ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
An Electrothermal Bipolar Tissue Sealing System (Ligasure) in Lung Surgery
Various diathermy techniques have been proposed to reduce intraoperative blood loss in surgery. In particular, standard bipolar technology is able to coagulate small vessels, with a diameter ranging from 1 mm to 3 mm, but it has some disadvantages, including sticking, charring, and significant thermal spread [1,2]. A new electrothermal bipolar tissue sealing system (LigaSure, Valleylab Inc., Boulder, Colorado, USA) recently has been applied in abdominal and pelvic surgery, mostly through laparoscopy [3-8]. The use of this sealing system allows the surgeon to improve vessel sealing with minimal thermal spread to the surrounding tissue. However, the potential role of this technique is still undefined and the experience in the literature is limited. We hereby report the technical characteristics of the system along with our initial experimental and clinical experience.
THE SYSTEM
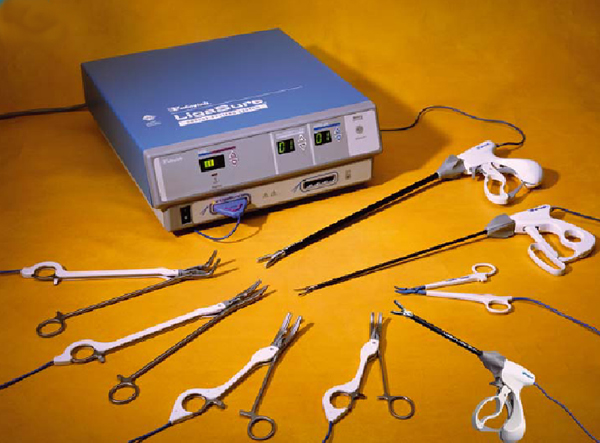
Figure 1. The complete “LigaSure Vessel Sealing System”, including an electrosurgical generator and several types of forceps.
The LigaSure Vessel Sealing System allows hemostasis by vessel compression and obliteration through the emission of bipolar energy. It includes (Figure 1):
1. An electrosurgical generator able to detect the characteristics of the tissue closed between the instrument jaws; it delivers the exact amount of energy needed to seal it permanently.
2. Several types of instruments that seal and, in some cases, divide the tissue. Those used in thoracic surgery are the following:
- LigaSure Atlas is a surgical endoscopic device (diameter: 10 mm, length: 37 cm) that seals and divides vessels up to 7 mm in diameter;
- LigaSure V is a single-use endoscopic instrument (diameter: 5 mm, length: 37 cm) able to seal and divide;
- LigaSure Lap is a single-use endoscopic instrument (diameter: 5 mm, length: 32 cm);
- LigaSure Precise is a single-use instrument (length: 16.5 cm) for open procedures specifically designed to provide permanent vessel occlusion to structures that require fine grasping;
- LigaSure Std is a reusable instrument;
HOW IT WORKS
This technique is different from the conventional coagulating methods that achieve vessel sealing by tissue carbonization. In fact, the heat generated from the bipolar energy determines the fusion of collagen and elastin in the walls of the vessel with the creation of a permanent sealed zone. The system detects the thickness of tissue to be coagulated and automatically defines the amount of energy required and the delivering time. An acoustic signal informs the surgeon when the vessel obliteration is complete and its division is possible. The seal zone shows a translucent appearance that is easy to recognize. Furthermore, this sealing system has a minimal thermal effect on the tissues surrounding the sealing line.
EXPERIMENTAL STUDIES
We evaluated the vessel and airways sealing capabilities of the LigaSure system in an ex vivo model with lungs of adult pigs, and analyzed the pathological effects and thermal damage of this technology on the surrounding lung tissue. We also evaluated the sealing capacity of this system on bronchi, vessels, and peripheral lung tissue, detecting the burst pressure for each anatomical structure. For bronchi, a pneumatic pressure of 60 mmHg, a value three-fold higher than the maximal physiologic airways pressure, was considered as the “critical pressure.” The percentage of LigaSure seal lines resistant to the critical pressure was 100% for 1-mm and 2-m vessels and it progressively decreased in the vessels with higher diameter. None of the bronchi with 6-mm or 7-mm diameter reached the critical pressure without opening. For blood vessels a hydrostatic pressure of 150 mmHg was considered as critical. For arteries and veins from 1 mm to 7 mm, the average burst pressure was higher than the critical pressure, and all the vascular suture lines remained intact to a pressure higher than 150 mmHg. For sealed lung margins a pneumatic pressure of 60 mmHg was considered critical. The average lung parenchyma seal burst pressure was higher than the critical pressure for all the groups; the percentage of sealed margins resistant to the critical pressure decreased slightly from 95% when a 0.2 gm wedge resection was performed to 68% when a 1.4 gm resection was performed (necessitating a thicker and longer sealed edge). Histological examination confirmed the sealing of vessels walls without necrotic coagulation of surrounding tissues. The depth of thermal injury at the seal line averaged 1.1 mm (range, 0.5 to 1.9 mm). The same findings were observed for bronchi with diameter ranging from 1 to 3 mm. For bronchi with higher sizes the seal obtained by the system was not satisfactory and the lumen often appeared patent at the level of the ligation line.
CLINICAL STUDIES
The LigaSure system was subsequently employed in 36 patients undergoing lung surgery. A total of 23 procedures were performed using LigaSure Standard forceps in 16 patients through thoracotomy (19 wedge resections, 2 segmentectomies, 2 fissure division; Video 1). Twenty patients underwent VATS using the LigaSure Atlas forceps handset, that has both vessel-sealing and tissue-dividing capabilitie (13 wedge resections, 5 bullectomies, 2 dissections of pleural adhesions). Video 2 shows a thoracoscopic bullectomy; the bulla appears completely adherent to the parietal pleura. In Video 3 the ablation of a nodule located on the diaphragmatic face of the lower lobe is shown. A number of parameters, including the presence of air leak at the completion of surgery, operating time, duration of thoracic drainage, and length of postoperative stay were recorded. The disease was benign in 25 patients, malignant in 11. The histology focused on the thermal effects of the sealing system on the lung tissue.
Video 1An Electrothermal Bipolar Tissue Sealing System (Ligasure) in Lung Surgery 2
Video 2
Video 3
In all patients the hemostasis obtained by LigaSure was effective, with minimal perioperative bleeding. There was no mortality. The mean operating time was 77.2 minutes (range, 60 to 97) for thoracotomies, and 60.3 minutes (range, 46 to 80) for thoracoscopies. The mean thoracic drainage duration was 3.1 days (range 1 to 8). Two patients had prolonged air leaks (>7 days) that resolved without requiring further intervention. No complications related to the use of this device were observed. The mean postoperative stay was 7.3 days (range 5 to 13) for thoracotomies and 4.6 days (range 1 to 6) for thoracoscopies. The histological evaluation of the surgical specimens showed minimal thermal spread in the tissues surrounding the sealing lines in all the cases.
DISCUSSION
The experience with the LigaSure system is limited, particularly in thoracic surgery [11-13]. Our study is the first biphasic evaluation of this tissue sealing system, both pre-clinical and clinical. The use of this coagulation technique in the swine model showed an excellent sealing effect on pulmonary vessels. All the vessels suture lines resisted to a pressure higher than 150 mmHg, which is above the pressures occurring in physiologic conditions. Thus, this technique can be safely used on vessels ≤7 mm in diameter, as reported in previous studies based on the use of the system in abdominal and gynecological surgery [3,5,6,8].
The effects of the sealing system on bronchi were not so satisfactory. Our results show that bronchi with diameter up to 2 mm in diameter, sealed by LigaSure, resisted to a pressure higher than 60 mmHg, that we conventionally considered as a “critical pressure”. The sealing effect was unpredictable for bronchi with diameter from 3 mm to 5 mm, and was negligible for bronchi with a diameter ≥6 mm. The good results obtained in smaller bronchi were confirmed by the assessment of suture margins after wedge resection performed using the system; the sealing capacity of lung tissue was adequate even with large size resections, reaching a weight of 1.4 g. These results indicate that LigaSure is a suitable system for wedge lung resection. They confirm the conclusions of a study by Tirabassi et al. [10] based on the swine survival model; these authors found that sealing of lung tissue by this system was similar to that by endoscopic staplers, and the burst strengths of lung resection margins obtained with both the methods was equal to that of normal lung tissue. As expected, our results suggest that this sealing system is not suitable for the closure of large bronchi, as is required in case of major lung resections.
The first case was reported was by Shigemura et al. [11]; afterward, the same research group reported the use of this sealing system in 12 video-assisted thoracoscopic procedures [12]. Albanese et al. [13] used successfully the system for thoracoscopic lobectomy in 14 small children with congenital lung lesions. We applied this coagulation technique either through thoracotomy or video-assisted thoracoscopy. In the latter procedures, the LigaSure Atlas forceps were used; they can be inserted through a 5 mm porthole, are easy to handle, and allow simultaneous coagulation and section of vessels and/or lung tissue. In our study, the use of this sealing technique improved the ablation of pleural adhesions and hemostasis during lung resections, reducing the use of conventional coagulation methods. We observed no perioperative major bleedings. Two patients undergoing bullectomy had prolonged air leaks that did not required additional procedures. The values of surgical parameters (operating time, length of postoperative drainage and hospitalization) were similar to those for conventional methods.
Compared to staplers, this system allows a better tailoring of nonanatomical lung resection margins, thus saving functional lung tissue, and avoids the use of multiple reloadable cartridges, reducing the costs of disposable surgical supplies. Another advantage is the minimal thermal spread to the tissues surrounding the sealing line; the pathological evaluation of the specimens obtained in both the phases of our study showed that the maximal depth of thermal damage was less than 2 mm (average value: 1.1 mm).
CONCLUSIONS
Pros
- It is easy to use and provides tissue sealing with permanent vessel fusion up to and including 7 mm in diameter without dissection or isolation [3,5,6,8]. Our study demonstrated that pulmonary vessel sealing lines withstand a pressure higher than 150 mmHg [14].
- The seal is natural, containing only the patient’s own collagen without foreign material.
- There is minimal sticking, tissue charring, and thermal spread, unlike standard bipolar and monopolar electrosurgery [9]. Our study showed that the maximal depth of thermal damage was less than 2 mm.
- The system is suitable for lysis of pleural adhesions, bullectomies, and lung wedge resections [10]; our results indicate that the sealing capacity of lung tissue is adequate even with large parenchymal resections.
- The device may be a valid alternative to staplers, particularly in the thoracoscopic setting [11-13].
- It avoids the use of multiple reloadable cartridges, thus reducing the costs of disposable surgical supplies.
Cons
- Initial cost.
- Learning curve
- The sealing effect of the system is unsatisfactory for bronchi with diameter >3 mm. Thus, Ligasure is not suitable for bronchial sutures and major lung resections.
- It is necessary to keep the jaws of the forceps clean in order to avoid the adhesion of the sealed tissue to the instrument.
References
- Memon MA. Surgical diathermy. Br J Hosp Med 1994;52:403-8.
- Spivak H, Richardson WS, Hunter JG. The use of bipolar cautery, laparosonic coagulating shears, and vascular clips for hemostasis of small and medium-sized vessels. Surg Endosc 1998;12:183-5.
- Kennedy JS, Stranahan PL, Taylar KD. High-burst strength, feedback-controlled bipolar vessel sealing. Surg Endosc 1998;12:867-868.
- Romano F, Caprotti R, Franciosi C, De Fina S, Colombo G, Uggeri F. Laparoscopic splenectomy using Ligasure. Preliminary experience. Surg Endosc 2002;16:1608-11.
- Shamiyeh A, Schrenk P, Tulipan L, Vattay P, Bogner S, Wayand W. A new bipolar feedback-controlled sealing system for closure of the cystic duct and artery. Surg Endosc 2002;16:812-3.
- Harold KL, Pollinger H, Matthews BD, Kercher KW, Sing RF, Heniford BT. Comparison of ultrasonic energy, bipolar thermal energy, and vascular clips for the hemostasis of small-, medium-, and large-sized arteries. Surg Endosc 2003;17:1228-30.
- Lee WJ, Chen TC, Lai IR, Wang W, Huang MT. Randomized clinical trial of Ligasure versus conventional surgery for extended gastric cancer resection. Br J Surg 2003;90:1493-6.
- Dubuc-Lissoir J. Use of a new energy-based vessel ligation device during laparoscopic gynecologic oncologic surgery. Surg Endosc 2003;17:466-8.
- Yim AP, Rendina EA, Hazelrigg SR, Chow LT, Lee TW, Wan S, Arifi AA. A new technological approach to nonanatomical pulmonary resection: saline enhanced thermal sealing. Ann Thorac Surg 2002;74:1671-6.
- Tirabassi MV, Banever GT, Tashjian DB, Moriarty KP. Quantitation of lung sealing in the survival swine model. J Pediatr Surg 2004;39:387-90.
- Shigemura N, Akashi A, Nakagiri T. New operative method for a giant bulla: sutureless and stapleless thoracoscopic surgery using the Ligasure system. Eur J Cardio-thorac Surg 2002;22:646-8.
- Shigemura N, Akashi A, Nakagiri T, Ohta M, Matsuda H. A new tissue-sealing technique using the Ligasure system for nonanatomical pulmonary resection: preliminary results of sutureless and stapleless thoracoscopic surgery. Ann Thorac Surg 2004;77:1415-19.
- Albanese CT, Sydorak RM, Tsao K, Lee H. Thoracoscopic lobectomy for prenatally diagnosed lung lesions. J Pediatr Surg 2003;38:553-5.
- Santini M, Vicidomini G, Baldi A, Gallo G, Laperuta P, Busiello L, Di Marino MP, and Pastore V. Use of an electrothermal bipolar tissue sealing system in lung surgery. Eur J Cardiothorac Surg 2006; 29: 226 - 230.




