ALERT!
This site is not optimized for Internet Explorer 8 (or older).
Please upgrade to a newer version of Internet Explorer or use an alternate browser such as Chrome or Firefox.
Necrotizing Esophagitis: The Black Esophagus
Introduction
Necrotizing esophagitis is a rare, potentially lethal clinical entity with an unclear etiology. We present a case of necrotizing esophagitis with a right-sided perforation of the distal esophagus in a 79 year-old female without significant precipitating events. The patient underwent resection of the distal two-thirds of the esophagus, followed by creation of a cervical esophagostomy, gastrostomy and feeding jejunostomy. The patient made an uneventful recovery and is awaiting reconstruction.
Case Presentation
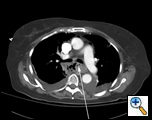
A 79 year-old female presented to an outside institution with a one day history of epigastric and retrosternal pain in the summer of 2005. She had no complaints of fever, emesis, hematemesis, retching, weight loss, odynophagia, or dysphagia. Her past medical history included hypothyroidism and stable coronary artery disease. She was thought to have acute cholecystitis, and seven days later developed respiratory distress and a chest X-Ray revealed mediastinal air and a right-sided pleural effusion. Computed tomography (CT) scan of the chest showed a large fluid collection around the distal esophagus tracking into the right chest, a possible intraluminal mass and air within the wall of the esophagus (Figure 1). A chest tube was placed in the right chest which drained culture-negative serosanguinous fluid. A gastrograffin swallow revealed an esophageal perforation with free-flow of contrast into the right chest (Figure 2). The patient was transferred to our institution with the diagnosis of esophageal perforation.
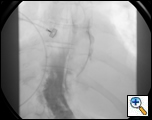
Figure 2: Gastrograffin swallow reveals free perforation of the esophagus with contrast flowing into the right chest.
Prior to transfer to our facility, the patient deteriorated clinically, was intubated and required vasopressors for blood pressure support. Upon arrival at our institution, her blood pressure was 110/70 mmHg on 3 mcg/kg/min of dopamine and 0.04 units/minute of vasopressin, pulse 100 beats per minute, oxygen saturation 98% on 40% fractional inspired O2. Her white blood cell count was 8.3k cells / mm3, hematocrit 26.7% and normal blood gas (pH 7.50, pO2 = 130, pCO2 = 31, base deficit = 1.5). Her admission electrolytes were (sodium = 138, potassium = 3.5, albumin = 2.3, creatinine = 0.6, chloride = 105, BUN = 7.0, glucose = 80). The patient was admitted to the intensive care unit and the decision made to take the patient to the operating room. Broad-spectrum antibiotics were continued (piperacillin-tazobactam) and an antifungal (fluconazole) was initiated.
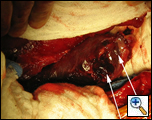
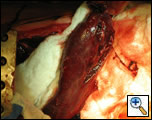
In the operating room, a right thoracotomy through the 5th intercostal space, 5th rib resection, and an intercostal muscle flap harvest was performed. Upon entering the chest, a large abscess was present surrounding the distal esophagus, with an empyema and widespread posterior mediastinitis. The esophagus exhibited full-thickness necrosis extending from the gastroesophageal junction to 5 cm below the thoracic inlet and was filled with clotted blood. There was a perforation of the distal esophagus extending from 10cm below the carina to the gastroesophageal junction (Figures 3A&B). The esophagus was resected with proximal and distal transections completed with an endoGIA 45 (3.5mm) stapling device. The posterior mediastinum was debrided and the right lung decorticated. Three chest tubes were placed, and a naso-gastric tube was placed within the cervical esophagus and both jejunostomy and gastrostomy tubes were placed. Forty eight hours later and off pressors, the patient was taken to the operating room for creation of a left cervical esophagostomy.
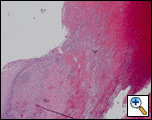
Figure 4: Histopathology reveals transmural acute and chronic inflammation with extensive ulceration and necrosis of the esophagus.
The patient made an uneventful postoperative recovery and was discharged from the hospital twelve days later. Surgical pathology demonstrated transmural acute and chronic inflammation with extensive ulceration and necrosis throughout the esophagus (Figure 4). Histology was negative for viral inclusions and malignancy. Microbiology revealed C. albicans from the mediastinal abscess.
Discussion:
Necrotizing esophagitis (NE) is a rare clinical entity with an unclear etiology. Causes include ischemia, trauma, caustic ingestion, radiation, and infection [1-4]. Ischemia is thought to occur in the elderly especially in those with cardiac and vascular disease and becomes manifest when the patient experiences a low flow state [5]. In the latter group, infectious esophagitis generally occurs in immunocompromised patients. Patients with acquired immune deficiency syndrome (AIDS) and hematologic malignancies make up the large majority of this group. Reports of NE secondary to infection are extremely uncommon in patients who are not immunocompromised. Mortality in NE is high, with overall rates approaching 50% [1-3]. In a retrospective review comparing surgical versus medical management, outcomes were superior in the patients who underwent surgical interventions. In fact, in this review mortality was 90% in the medically managed group versus 27% in the surgical group [3].
NE is often discovered on upper endoscopy, and has been described as “black esophagus” in this setting. Conservative management with antibiotics, fluid resuscitation, nutritional support and proton pump inhibitors are often effective with surgical intervention required when patients fail this therapy. Transmural involvement is also exceedingly rare, with most patients presenting with mucosal and submucosal involvement. Frank perforation is an atypical presentation when the esophagitis is discovered on endoscopy [1,4].
Our patient presents with NE from an unclear source. It may be that low flow/ischemia contributed to the development of disease in this patient with a history of coronary artery disease. However, acute coronary syndrome was ruled out on admission.
The presence of intramural air may be a clue in the early diagnosis of this condition in the absence of overt clinical findings. In a patient with clinical findings early operative intervention improves survival. Removal of the source of sepsis and inflammation, i.e., the esophagus, drainage of the mediastinum, chest and gastrointestinal tract, appropriate antibiotic and antifungal coverage, and nutritional support are vital to a successful outcome.
References
1. Augusto F, Fernandes V, Cremers MI, et al. Acute necrotizing esophagitis: a large retrospective case series. Endoscopy 2004;36:411-5.
2. Gaissert HA, Breuer CK, Weissburg A, and Mermel L. Surgical management of necrotizing Candida esophagitis. Ann Thorac Surg 1999;67:231-3.
3. Gaissert HA, Roper CL, Patterson GA, and Grillo HC. Infectious necrotizing esophagitis: outcome after medical and surgical intervention. Ann Thorac Surg 2003;75:342-7.
4. Obermeyer R, Kasirajan K, Erzurum V, and Chung D. Necrotizing esophagitis presenting as a black esophagus. Surg Endosc 1998;12:1430-3.
5. Pantanowitz L, Gelrud A, and Nasser I. Black esophagus. Ear Nose Throat J 2003;82:450-2.